Sarcopterygii: Osteolepiformes: Tristichopteridae (2)
Cabonnichthys & Mandageria
Taxa on This Page
-
Cabonnichthys X
-
Mandageria X
Demorphing the Tetrapodomorpha: 5. The Axial Skeleton
Reversing Polarities in the Spine
The rhipidistians appear to have developed two types of axial skeleton. If current phylogenies are approximately correct, these morphs do not appear to represent the expected evolutionary, or perhaps even ecological, succession. Interestingly, both arrangements are observed in tetrapods. The first is the rachitomous vertebra. Many early tetrapods have this type of vertebra. The centrum is dominated by the large ventral elements, the intercentra. These are often joined or fused at the ventral midline and extend around the notochord in both directions, ending at, or just short of, the neural arches. The dorsal elements, the pleurocentra are paired, not fused, and articulate fairly loosely with the neural arches and intercentra. Osteolepis has this kind of vertebra. The other morph is the holospondylous ring-like vertebra, similar to the vertebra of living amphibians. Here, the notochord is completely surrounded by a ring of bone which may represent either the intercentra, or fused intercentra plus pleurocentra. The neural arch sits on top of the ring like the gem setting on a wedding ring. Megalichthys has this type of vertebra.
Andrews & Westoll (1970a) discuss a supposed transformation series in the spine from the loosely bound elements of Eusthenopteron, with small pleurocentrum, through Osteolepis large pleurocentrum, more closely bound), to Megalichthys (complete fusion). Unfortunately, that series is inconsistent with current views of sarcopterygian phylogeny. They argue that the arrangements in Eusthenopteron and Osteolepis are morphologically similar to rachitomous vertebra, but are mechanically distinct because of the larger notochord. They argue that this vertebral type would add mechanical strength to the spine, but would permit permitting substantial torque and flexure. The fused Megalichthys form would not permit torque, tending to hold the body straight while permitting thunniform?) swimming by lateral flexure. The unspoken assumption seems to be that these mechanical restriction would perform better in terrestrial locomotion. Andrews & Westoll observe that this type of vertebra was very common in Carboniferous forms including Dipnoi, rhizodonts, and embolomeres. However, the known distribution suggests that, if anything, the ring-like vertebra is primitive for Rhipidistia. The rachitomous vertebra may be a synapomorphy of the clade Tristichopteridae + Elpistostegalia. Its difficult to tell, since the vertebrae of the elpistostegalian fishes are not well known.
The tristichopterids and elpistostegalians do seem to share another important axial novelty -- the beginnings of the tetrapod rib. The structure is well-understood only in Eusthenopteron. The mid-trunk vertebrae of Eusthenopteron have a well-defined rib with a capitulum articulating with a parapophysis at mid-height on the intercentrum and an unfinished dorsal end (tuberculum) which presumably had a cartilaginous connection with the adjacent neural arch. The shaft of the rib is minimal and is directed dorsolaterally and posteriorly. Clearly, the principle function was to stiffen the spine, rather than the body wall. Panderichthys may have had well-developed rib shafts, possibly even enlarged ribs (as in Ichthyostega); but their structure is not certain.
 The axial skeleton does exhibit one fairly well-established trend. The body becomes variably longer and the anal, pelvic and second dorsal fins tend to diminish in size and move posteriorly, sometimes merging with the tail fin. The tail fin develops a progressively lower aspect ratio. Ahlberg & Johanson (1997). This tendency is strongly developed in the tristichopterids, probably true of the elpistostegalians and dipnomorphs, and perhaps even applicable to the rhizodonts. In fact, we can add that the skull also becomes longer and flatter, with the orbits somewhat dorsally placed. The latter characters are strongly present in the Elpistostegalia, reasonably so in tristichopterids and rhizodonts, and is probably not applicable to dipnoans. These concerted changes have prompted several authors to speculate that these rhipidistian groups were all converging on an ambush predator lifestyle, because of the similarity to Esox, the extant pike. Compare the image of Esox at right to the reconstruction of Cabonnichthys below.
The axial skeleton does exhibit one fairly well-established trend. The body becomes variably longer and the anal, pelvic and second dorsal fins tend to diminish in size and move posteriorly, sometimes merging with the tail fin. The tail fin develops a progressively lower aspect ratio. Ahlberg & Johanson (1997). This tendency is strongly developed in the tristichopterids, probably true of the elpistostegalians and dipnomorphs, and perhaps even applicable to the rhizodonts. In fact, we can add that the skull also becomes longer and flatter, with the orbits somewhat dorsally placed. The latter characters are strongly present in the Elpistostegalia, reasonably so in tristichopterids and rhizodonts, and is probably not applicable to dipnoans. These concerted changes have prompted several authors to speculate that these rhipidistian groups were all converging on an ambush predator lifestyle, because of the similarity to Esox, the extant pike. Compare the image of Esox at right to the reconstruction of Cabonnichthys below.
We disagree -- at least in part. The two body forms differ in some critical details. Esox has the following important characters absent from rhipidistians: caudal fin dorsoventrally tall and C-shaped, long caudal peduncle, anal and dorsal fin of moderate size, located just anterior to the peduncle, pelvic fins almost at the middle of the body, and all paired fins relatively small. These characters are associated with an adaptation for very high acceleration using the tail, with the attitude of the long body adjusted by the fins located at certain critical junctures: at the break between the rapidly moving tail and the body, near the center of gravity, and just behind the head. the size and shape of the caudal (and, to a lesser degree, the dorsal & anal fins) is important, since the C-shaped tail not only provides a large surface area, but tends to shed turbulent gyres well away from the body and off to the side. Turbulence behind the animal creates drag. Turbulence off to the side and at some distance doesn't slow the fish down as badly. The sharply distinct peduncle is necessary to damp the tendency of the lashing tail to throw off the carefully adjusted aim of the body towards the prey animal.
Rhipidistians are clearly designed for something else. The small, rhomboid tail would not sweep much water in its arc, and turbulent gyres would tend to be shed directly behind the animal from the tip of the middle lobe. Rhipidistians have neither a distinct caudal peduncle [2], nor large posterior fins to stop the spread of the tail oscillation into the trunk of the body. The pelvic fins are small and far behind the center of gravity, while the pectoral fins are too large to minimize frictional drag.
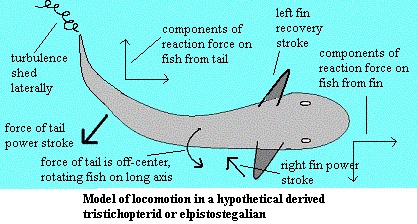 What this may be instead is the beginning of a body adapted for a tetrapod locomotor style -- actually a bipedal style in these fishes. The peduncle is absent precisely in order to twist the body slightly. in the image, the caudal fin is moving toward the centerline in its power stroke. Osteolepiforms are somewhat bottom heavy.Since the force of the tail is applied somewhat above the center of gravity, (see image) the body rotates to the right. As the left side of the body rises, the left pectoral fin rises naturally in a recovery stroke. The same rotation helps the right fin work extend to begin its power stroke. As the body rocks back toward balance, the pectoral fin adductors dig in to the water, below the center of gravity, counteracting the tortion of the tail. The lateral components of the reaction forces from tail and fin also tend to cancel each other out. However, the fin and tail power strokes might be asynchronous, with the fin reaching maximum power output as the tail passes the centerline, and into its recovery stroke. The result may have been a remarkably smooth motion, like the crawl performed by an expert swimmer. [1]
What this may be instead is the beginning of a body adapted for a tetrapod locomotor style -- actually a bipedal style in these fishes. The peduncle is absent precisely in order to twist the body slightly. in the image, the caudal fin is moving toward the centerline in its power stroke. Osteolepiforms are somewhat bottom heavy.Since the force of the tail is applied somewhat above the center of gravity, (see image) the body rotates to the right. As the left side of the body rises, the left pectoral fin rises naturally in a recovery stroke. The same rotation helps the right fin work extend to begin its power stroke. As the body rocks back toward balance, the pectoral fin adductors dig in to the water, below the center of gravity, counteracting the tortion of the tail. The lateral components of the reaction forces from tail and fin also tend to cancel each other out. However, the fin and tail power strokes might be asynchronous, with the fin reaching maximum power output as the tail passes the centerline, and into its recovery stroke. The result may have been a remarkably smooth motion, like the crawl performed by an expert swimmer. [1]
Actually, a fish along these lines might have adopted several possible gaits. The illustrated cycle is but one example. Further, if the fish were large and long enough -- perhaps the size and proportions of Acanthostega -- it would be possible to shift much of the propulsive function to hind legs. The advantage of doing so is that the motion of the legs would offset the torque generated by the tail, leaving the anterior body free to steer, generate additional propulsion, or maneuver the mouth over some prospective menu item, as circumstances permitted.
Frank Fish (1998) has pointed out that locomotion is very conservative. Fish's Law (our phrase) is that changes in locomotor pattern lag changes in lifestyle. It is "easier" in an evolutionary sense (more probable, less complex) to change body forms and maintain an ancestral locomotor pattern than to rewire the brain and muscles to adopt an entirely new system of movement. If Fish's Law applies here -- and it should -- we should reasonably expect osteolepiforms to evolve a style of aquatic locomotion which looks like walking. The mechanical analysis above suggests that this is exactly what occurred. ATW031107.
[1] Note also that, if a substantial part of the trunk is involved in the motion of the tail, then two mechanical consequences follow. First, the caudal fin doesn't need to be so large, because a large body segment is exerting force on the water, not just the caudal fin. Second, the tip of the tail will be rotated laterally by a wide angle, so that turbulent gyres will be shed laterally, reducing drag.
[2] Osteologically, these forms have a large peduncle. However, the peduncle is never reconstructed to be mechanically distinct as it is in Esox.
 Cabonnichthys & Mandageria
Cabonnichthys & Mandageria
Cabonnichthys and Mandageria are two tristichopterids recently described from the Canowindra assemblage. Both have an odd, conical body form with a rhomboid tail. All of the fins are at least somewhat reduced except the pectoral fins. The significance of this design has been discussed above.
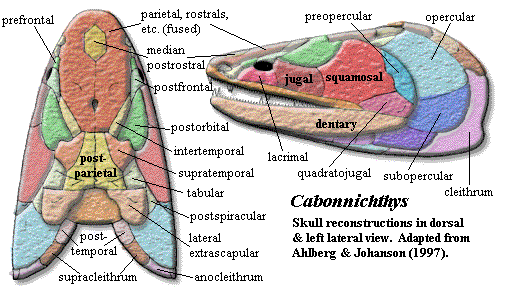
Cabonnichthys is, in some ways, a throwback. The snout is relatively short and blunt. The rostral bones are small, indistinct and superficially fused. Cabonnichthys lacks any trace of a separate frontal bone. The parietal is much larger than the postparietal, and the snout bears a single median rostral. The jugal not only participates in the orbit, but contacts the postfrontal and excludes the postorbital from the orbit -- a probable apomorphy of the genus. The dermal intracranial joint seems well-developed. However, the skull table is oddly constricted at this point, and it is unclear whether the joint would operate in the normal way.
In other aspects, Cabonnichthys appears derived. The parietal foramen is close to the posterior margin of the parietal and well behind the orbits. There is a relatively sharp break between skull table and cheek. The postorbital and squamosal are large. They meet and may underlie the skull table bones, rather than being sutured to them. The supratemporal has a "horn" that fits into a notch in the postorbital, and the lateral extrascapulars also appear to have a complex, interlocking arrangement with surrounding bones.
The pectoral girdle is very robust. The scapulocoracoid is still tripodal, but it is massive and the large foramina present in Eusthenopteron are almost closed. The "humeral process" is absent, and the ectepicondyle has a more tetrapod-like distal location and is continuous with the ulnar facet. The scales lack any cosmine and are ornamented with anteroposteriorly oriented grooves. While this feature is not noted by Ahlberg & Johanson (1997), the opercular bones seem to be reduced.
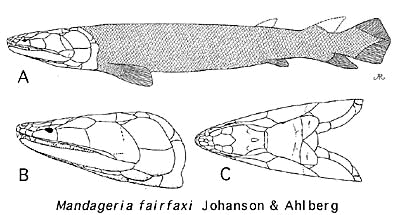 The most noticeable thing about Mandageria is that it is huge. At nearly 2m, Mandageria is twice as long as Gooloogongia, the second largest fish in the Canowindra fauna. Like Cabonnichthys, it has some startlingly primitive features of the rostrum, but -- unlike Cabonnichthys-- the rostrum is long and pointed. The skull is quite flat. The dermal intracranial joint is not only clear, but marked by a distinct parietal-postparietal gap bridged by small dermal bones without fixed identity.
The most noticeable thing about Mandageria is that it is huge. At nearly 2m, Mandageria is twice as long as Gooloogongia, the second largest fish in the Canowindra fauna. Like Cabonnichthys, it has some startlingly primitive features of the rostrum, but -- unlike Cabonnichthys-- the rostrum is long and pointed. The skull is quite flat. The dermal intracranial joint is not only clear, but marked by a distinct parietal-postparietal gap bridged by small dermal bones without fixed identity.
The most remarkable feature of Mandageria is that it had developed a structure somewhat analogous to the tetrapod neck. We have mentioned this structure in a previous section (see image). In essence, the ventral fontanelles have been expanded, and the exoccipital has developed paired articular surfaces contacting neural arches of the first vertebra. It is not clear how this joint functioned with the pectoral girdle still anchored to the dermal skull. Presumably there was enough play in the long posterior skull to allow some degree of bending.
These two Australian forms are probably only the visible tip of a large hidden diversity in Late Devonian tristichopterids. Derived rhipidistians of all sorts show mosaic convergence on the tetrapod suite of characters. What is unclear is whether the tetrapods were the result of successful adaptation, or simply a peculiar branch of a general, broad radiation of rhipidistian forms into a novel environment. ATW031107.
Descriptions
 Cabonnichthys: C. burnsi Ahlberg & Johanson, 1997.
Cabonnichthys: C. burnsi Ahlberg & Johanson, 1997.
Range: Famennian of Australia (NSW) [AJ97].
Phylogeny: Tristichopteridae:: Mandageria + *.
Characters: ~75 cm [AJ97]; head broad & short, with blunt snout [AJ97]; snout bones largely fused & indistinct externally [AJ97]; premaxilla always superficially fused with rostrals [AJ97]; maxilla posterodorsal process low [AJ97]; ~4 nasals and 1 large median postrostral [AJ97]; no distinct frontals; prefrontal contacts tectal (which?) [AJ97]; prefrontal does not contact lateral rostral [AJ97] [1]; parietals 1.3X as long as postparietals [AJ97]; pineal foramen close to posterior margin of parietal [AJ97$]; pineal bones kite-shaped, with distinct posterior (= distal?) point [AJ97$]; tabular & supratemporal fuse to postparietals in older specimens [AJ97]; intertemporal contacts postfrontal [AJ97$]; supratemporal with small lateral "horn" fitting into notch in postorbital [AJ97$]; small, crescentic postspiracular [AJ97]; orbits small, anterior & somewhat dorsal [AJ97]; postorbital long & flares posteriorly, contacting intertemporal, squamosal & jugal [AJ97]; postorbital even broader internally & underlies parietal extensively [AJ97]; jugal contacts postfrontal & excludes postorbital from orbit [AJ97$]; jugal rectangular [AJ97]; squamosal meets but does not suture with supratemporal, tabular & postspiracular [AJ97]; palate very similar to Mandageria, but wider [AJ97]; parasphenoid quite broad [AJ97]; accessory vomers large [AJ97]; premaxilla with enlarged tooth at anterior end [AJ97$]; palatine & ectopterygoid with complete marginal tooth rows [AJ97]; parotic plate present [AJ97]; parasymphysial fang pair present on small plate [JA97] [AJ97$]; dentary long & slender, with single row of small teeth [AJ97]; coronoids without marginal teeth [AJ97$] (or [JA97]: limited to most posterior part of posterior coronoid); coronoids separated by deep fossa [AJ97]; prearticular very large, suturing to splenial anteriorly [AJ97]; caudal fin homocercal & diamond-shaped [AJ97$]; caudal fin with deep peduncle & low aspect ratio, with both lobes supported by independent radials articulating with neural/hemal arches [AJ97]; lateral extrascapulars triangular and slightly indented medially [AJ97$]; pectoral girdle generally as in Eusthenopteron [AJ97]; scapulocoracoid very robust, tripodal but with small openings [AJ97]; glenoid large, with widest part proximal to cleithrum (opposite of Eusthenopteron) [AJ97]; anterior of infra- & supraglenoid buttresses unfinished & possibly joined with cartilage [AJ97]; pectoral fin as in Eusthenopteron but without posterolateral corner [AJ97]; humeral process probably absent [AJ97]; ectepicondyle reaches distal end of humerus & may be continuous with ulnar facet [AJ97]; scales without cosmine & with teardrop boss on internal surface, ornamented with anteroposterior grooves [AJ97].
Notes: [1] This is a bit unclear. There seems to be considerable individual variation, the bones are variably fused, and none of the specimens are undamaged in this region.
Image: Cabonnichthys from the Age of Fishes Museum
Links: Cabonnichthys burnsi; ^Palaeontology - Cabonnichthys burnsi Ahlberg & Johanson, 1997; Second tristichopterid (Sarcopterygii, Osteolepiformes) from the ...; ^Palaeontology - Canowindra - Cabonnichthys burnsi; ^Palaeontology - Canowindra - Cabonnichthys burnsi reconstruction.
References: Ahlberg & Johanson (1997) [AJ97], Johanson & Ahlberg (1997) [JA97]. ATW031019.
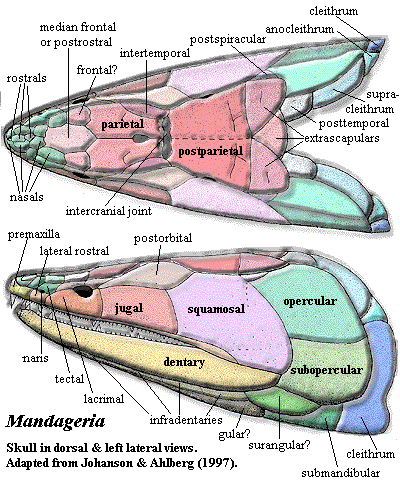 Mandageria: M. fairfaxi Johanson & Ahlberg, 1997.
Mandageria: M. fairfaxi Johanson & Ahlberg, 1997.
Range: Frasnian of Australia (originally reported as Famennian in [JA97], but Frasnian in [J+03]).
Phylogeny: Tristichopteridae:: Cabonnichthys + *.
Characters: 1.5-2.0 m; head narrow & acutely pointed [JA97]; premaxilla deep anteriorly [JA97]; anterior nasal separates premaxilla from anterior rostral [JA97]; lateral rostral may be two separate bones [JA97]; premaxilla probably has no external exposure at level of naris [JA97]; parietals small, closely sutured at midline [JA97]; pineal small, posterior & covered by series of small bones [JA97]; postfrontal fails to contact intertemporal [JA97]; parietal - postparietal suture located far behind orbits [JA97]; space between parietal & postparietal filled with small anamestic bones [JA97]; extratemporal absent [JA97]; postparietals, tabulars & supratemporals fused externally [JA97]; lateral extrascapulars triangular, overlap median extrascapular extensively & almost meet in midline anteriorly [JA97]; postspiracular bone located between lateral extrascapulars and opercular [JA97]; orbits small, lateral & located in anterior third of skull [JA97] [J+03]; postorbital elongate & very narrow anteriorly [JA97]; jugal & postorbital excluded from orbit by postfrontal & strongly developed lacrimal [JA97]; postfrontal with elongate posterior corner, but does not contact intertemporal [JA97]; jugal rectangular & larger than lacrimal [JA97]; quadratojugal area is not known & quadratojugal may be present [JA97]; preopercular also unknown but almost certainly present [JA97]; operculogular series standard, with opercular, subopercular, ~7 submandibulars, lateral (=principal) gulars and small median gular plate [JA97]; exoccipital with paired articular surfaces contacting neural arches of atlas, creating functional "neck" [J+03]; basioccipital probably absent or unossified [J+03]; hyomandibular does not reach jaw articulation [J+03]; hyomandibular strongly twisted [J+03]; ventral hyomandibular articulation located much more posteriorly than anterior (see image) [JA97]; hyomandibular articulations located about middle of vestibular fontanelle due to anterior extension of fontanelle [J+03]; ventral hyomandibular articulation displaced posterior to dorsal articulation [J+03]; lateral commisure triangular, broad ventrally, with ventral flange wrapping around ventral hyomandibular articulation [J+03]; lateral commisure with sharp ridge on anterior margin [J+03]; large tooth (not fang pair) on an ethmoid plate within apical fossa perimedian position [J+03] [2]; vomers with fang pairs & row of small teeth [JA97]; parasphenoid denticulated plate wider & shorter than in Eusthenopteron [JA97]; parasphenoid denticulate plate flanked by "accessory vomers" [JA97]; pterygoid short, with palatal exposure barely reaching vomers [JA97]; parasymphysial fang pair present [JA97]; parasymphysial plate small, not contacting coronoid [JA97]; median premaxillary tooth close to tooth row, which is broken at the premaxillary symphysis [JA97]; deep precoronoid fossa lingual to anterior dentary traversed by rib from splenial and anterior of prearticular which supports parasymphysial plate [JA97]; coronoid marginal teeth limited to most posterior part of posterior coronoid [JA97]; branchial apparatus (image) as in Eusthenopteron, with denticulated plates [JA97] [3]; vertebrae poorly known, probably like Eusthenopteron [JA97]; ribs very small [J+03]; caudal fin diphyceral with hypochordal lobe slightly larger [JA97]; caudal fin with low aspect ratio [JA97] [4]; typical shoulder girdle, with posttemporal,  supracleithrum, anocleithrum & cleithrum [JA97] shoulder girdle loosely constructed & loosely attached to skull [J+03]; interclavicle uncertain [JA97]; supracleithrum elongate [JA97]; scapulocoracoid probably tripodal [JA97]; pectoral fin exactly as in Eusthenopteron except for details of humerus [JA97]; humerus not dorsoventrally flattened [J+03]; entepicondyle opens at anterior base of epicondyle and exits on distal surface between epicondyle and ulnar articulation [JA97]; notch in entepicondyle, gripping edge of ulna [JA97]; humerus with ventral ridge as in Eusthenopteron [JA97]; ectepicondyle large and flat [JA97]; deltoid process present [JA97]; radius with uniquely thick, coarsely ornamented scales [JA97]; scales very small & numerous [JA97]; scales with median internal boss [JA97]; scales without cosmine, and with ornament of fine anteroposterior lines [JA97]; mid-water fish, not land dweller or benthic [J+03].
supracleithrum, anocleithrum & cleithrum [JA97] shoulder girdle loosely constructed & loosely attached to skull [J+03]; interclavicle uncertain [JA97]; supracleithrum elongate [JA97]; scapulocoracoid probably tripodal [JA97]; pectoral fin exactly as in Eusthenopteron except for details of humerus [JA97]; humerus not dorsoventrally flattened [J+03]; entepicondyle opens at anterior base of epicondyle and exits on distal surface between epicondyle and ulnar articulation [JA97]; notch in entepicondyle, gripping edge of ulna [JA97]; humerus with ventral ridge as in Eusthenopteron [JA97]; ectepicondyle large and flat [JA97]; deltoid process present [JA97]; radius with uniquely thick, coarsely ornamented scales [JA97]; scales very small & numerous [JA97]; scales with median internal boss [JA97]; scales without cosmine, and with ornament of fine anteroposterior lines [JA97]; mid-water fish, not land dweller or benthic [J+03].
Note: [1] the diagnosis states "postrostral," and this word is also used inconsistently in the text. It appears to be a typographical error. [2] similar structures are found in megalichthyids, certain other tristichopterids, at least some Rhizodonts and (without a tooth) in Acanthostega and Crassigyrinus, but these are premaxillary processes into the apical vacuity [JA97]. [3] Johanson & Ahlberg [JA97] find that the hypobranchial of the 4th branchial arch attaches to the 3rd hypobranchial, rather than to a basibranchial. They find no evidence for a
5th branchial arch. They argue, very persuasively, that this is the general condition for all Rhipidistians, including Eusthenopteron. [4] Mandageria and similar fishes have been repeatedly compared to Esox in probable habits and mode of life.
Image: Body reconstruction: Mandageria from the Australian Museum Online. See also image of branchial apparatus at Rhipidistia. Pectoral fin reconstruction: based on two images in [JA97], eked out with Eusthenopteron. Interestingly, it would require doing considerable violence to the known components to create a fin with most of the lepidotrichia on the preaxial side as in Eusthenopteron. The most natural arrangement puts the lepidotrichia on the distal end in a tetrapod-like position. However, this is our own reconstruction -- not one which can fairly be ascribed to Johanson & Ahlberg -- and is not reliable.
Links: Mandageria fairfaxi; Palaeontology - Canowindra slide show; Annual Conference Abstracts 2001 Ahlberg abstract).
References: Johanson & Ahlberg (1997) [JA97]; Johanson et al. (2003) [J+03] ATW031009.
 The axial skeleton does exhibit one fairly well-established trend. The body becomes variably longer and the anal, pelvic and second dorsal fins tend to diminish in size and move posteriorly, sometimes merging with the tail fin. The tail fin develops a progressively lower aspect ratio. Ahlberg & Johanson (1997). This tendency is strongly developed in the tristichopterids, probably true of the elpistostegalians and dipnomorphs, and perhaps even applicable to the rhizodonts. In fact, we can add that the skull also becomes longer and flatter, with the orbits somewhat dorsally placed. The latter characters are strongly present in the Elpistostegalia, reasonably so in tristichopterids and rhizodonts, and is probably not applicable to dipnoans. These concerted changes have prompted several authors to speculate that these rhipidistian groups were all converging on an ambush predator lifestyle, because of the similarity to Esox, the extant pike. Compare the image of Esox at right to the reconstruction of Cabonnichthys below.
The axial skeleton does exhibit one fairly well-established trend. The body becomes variably longer and the anal, pelvic and second dorsal fins tend to diminish in size and move posteriorly, sometimes merging with the tail fin. The tail fin develops a progressively lower aspect ratio. Ahlberg & Johanson (1997). This tendency is strongly developed in the tristichopterids, probably true of the elpistostegalians and dipnomorphs, and perhaps even applicable to the rhizodonts. In fact, we can add that the skull also becomes longer and flatter, with the orbits somewhat dorsally placed. The latter characters are strongly present in the Elpistostegalia, reasonably so in tristichopterids and rhizodonts, and is probably not applicable to dipnoans. These concerted changes have prompted several authors to speculate that these rhipidistian groups were all converging on an ambush predator lifestyle, because of the similarity to Esox, the extant pike. Compare the image of Esox at right to the reconstruction of Cabonnichthys below. What this may be instead is the beginning of a body adapted for a tetrapod locomotor style -- actually a bipedal style in these fishes. The peduncle is absent precisely in order to twist the body slightly. in the image, the caudal fin is moving toward the centerline in its power stroke. Osteolepiforms are somewhat bottom heavy.Since the force of the tail is applied somewhat above the center of gravity, (see
What this may be instead is the beginning of a body adapted for a tetrapod locomotor style -- actually a bipedal style in these fishes. The peduncle is absent precisely in order to twist the body slightly. in the image, the caudal fin is moving toward the centerline in its power stroke. Osteolepiforms are somewhat bottom heavy.Since the force of the tail is applied somewhat above the center of gravity, (see  Cabonnichthys & Mandageria
Cabonnichthys & Mandageria


 supracleithrum, anocleithrum & cleithrum [JA97] shoulder girdle loosely constructed & loosely attached to skull [J+03]; interclavicle uncertain [JA97]; supracleithrum elongate [JA97]; scapulocoracoid probably tripodal [JA97]; pectoral fin exactly as in Eusthenopteron except for details of humerus [JA97]; humerus not dorsoventrally flattened [J+03]; entepicondyle opens at anterior base of epicondyle and exits on distal surface between epicondyle and ulnar articulation [JA97]; notch in entepicondyle, gripping edge of ulna [JA97]; humerus with ventral ridge as in Eusthenopteron [JA97]; ectepicondyle large and flat [JA97]; deltoid process present [JA97]; radius with uniquely thick, coarsely ornamented scales [JA97]; scales very small & numerous [JA97]; scales with median internal boss [JA97]; scales without cosmine, and with ornament of fine anteroposterior lines [JA97]; mid-water fish, not land dweller or benthic [J+03].
supracleithrum, anocleithrum & cleithrum [JA97] shoulder girdle loosely constructed & loosely attached to skull [J+03]; interclavicle uncertain [JA97]; supracleithrum elongate [JA97]; scapulocoracoid probably tripodal [JA97]; pectoral fin exactly as in Eusthenopteron except for details of humerus [JA97]; humerus not dorsoventrally flattened [J+03]; entepicondyle opens at anterior base of epicondyle and exits on distal surface between epicondyle and ulnar articulation [JA97]; notch in entepicondyle, gripping edge of ulna [JA97]; humerus with ventral ridge as in Eusthenopteron [JA97]; ectepicondyle large and flat [JA97]; deltoid process present [JA97]; radius with uniquely thick, coarsely ornamented scales [JA97]; scales very small & numerous [JA97]; scales with median internal boss [JA97]; scales without cosmine, and with ornament of fine anteroposterior lines [JA97]; mid-water fish, not land dweller or benthic [J+03].