
| Neopterygii | ||
| The Vertebrates | Overview |
| Vertebrates Home | Vertebrate | Vertebrate |
|
Abbreviated Dendrogram
Teleostomi
├─Sarcopterygii
└─Neopterygii
├─Ginglymodi
│ ├─Macrosemiiformes
│ └─Lepisosteiformes
└─Halecostomi
├─Halecomorphi
│ ├─Parasemionotidae
│ └─Amiiformes
└─┬─┬─Semionotiformes
│ └─Pycnodontiformes
└─┬─Pachycormiformes
└─Teleostei
|
Contents
Overview |
 |
 |
 |
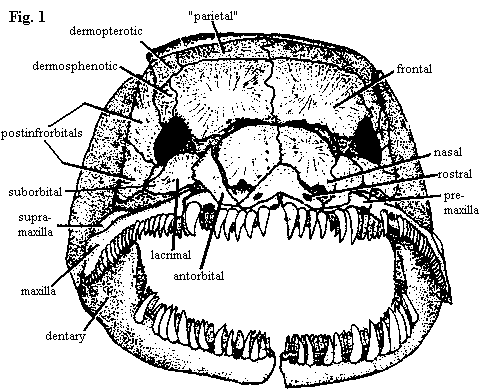
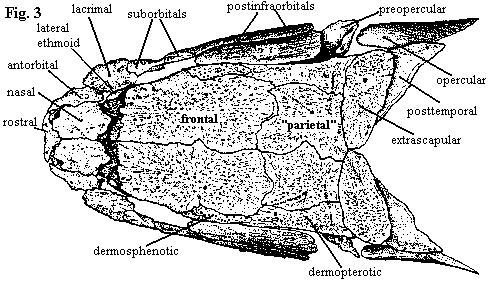
| Figures 1-3. Amia calva: skull in anterior, left lateral, and dorsal views. Branchiostegal rays have been removed. Adapted from Grande & Bemis (1998). |
"It is always met with in fresh water, and is an ugly, ill shaped and ill looked and sluggish fish; of little use for the table."
-- Alexander Garden (1763), in a letter to Linnaeus, enclosing the dried holotype of Amia calva.
Grande & Bemis (1998) is a vast storehouse of neopterygian knowledge, small samplings of which will be digested, repackaged, and doled out at different points in this section. Perhaps the longest single portion of the work is G&B's redescription of the living bowfin, Amia calva Linnaeus 1766. With apologies to G&B, Garden's original description, quoted above, is no more than the truth. However, Amia is one of the very few non-teleost neopterygians still living. It is the only such fish which comes close to our mental morphotype of what constitutes a "fish" without actually having most of the awkward teleost specializations. Thus, if we can but lay aside Garden's aesthetic objections, Amia makes an excellent study for understanding the basic makeup of actinopterygians, as well as providing us with essential background to place the teleost radiation in its proper context. While it is not really possible summarize G&B's description in a few hundred words, we can at least hit the high spots of the skull. The rest of the tail will have to deferred.
The premaxillae (fig. 1) form a slightly arched lateral bar across the front of the rostrum. Posterodorsally, they bear long, broad processes which enclose large, tear-drop shaped nares. However, this dorsal part of the premaxilla is covered by the nasals, rostrals and antorbitals. Oddly, the tooth-bearing bar and narial process begin as separate ossifications, so that we may question whether the dorsal process is actually homologous with the premaxilla in other vertebrates, or whether it bears a closer relationship to the septomaxilla. Note that the rostral has two ventrolateral "wings."
The maxilla itself (figs. 2 & 8) does not contact the premaxilla. It looks quite a lot like a teleost maxilla, complete with a single supramaxilla (teleosts have more). However, its primary mobility is lateral. The anterior end articulates with a socket formed by the pre-ethmoids, and the posterior ends are not fixed to the cheek, allowing the posterior maxilla to swing outward. The maxilla bears a conspicuous notch at its posterior end.
The nasals (fig. 3) are relatively small, plate-like and do not contact the frontals directly. The gap between nasals and frontals presumably permits the maxilla and premaxilla to rotate with respect to each other. The frontals are the largest of the skull roofing bones. They are occasionally fused across the midline. In fact all of the roofing bones, including the dermopterotics, are individually variable and may fuse to neighboring bones. The frontal gives the appearance of perhaps being doubled, and, in fact, it probably includes the parietal, as that term is used in tetrapod osteology. Note, for example, that the pineal organ lies under the posterior half of the frontal (Amia has no parietal foramen). Thus the "parietals" of most actinopterygians are probably homologous with the postparietals of tetrapods. These bones, whatever their true nature, are notable for extending thin lamina under the surface of the adjacent dermal bones, thus presumably adding stability to the apex of the skull roof. The homology of the posttemporals is unclear.
In adults of Amia, the dermosphenotics and dermopterotics are also consolidated with the skull roof. This is not the case in all actinopterygians, or even all neopterygians. The dermopterotic is actually formed by the fusion of an anterior and posterior canal-bearing bones. As with many of the other dermal bones, particularly the circumorbital series, these begin life as simple tubes enclosing a sensory line. The dermopterotic is also notable for forming the dorsal articulation of the hyomandibular. These bones are only loosely associated with the postinfraorbitals below them.
On the lateral skull (fig. 2), the circumorbital bones are unusually tidy and well-organized. Rather than being scattered around more or less at random, they are disposed in tidy pairs about the orbit -- more like a formal dance than a beer party. The pair posterior to the orbit, which Grande & Bemis perversely insist on calling postinfraorbitals, are elongate, filling the long gap between the orbit and the hyomandibular and overlapping most of the dermosphenotic. By contrast, the two suborbitals G&B usually refer to them as subinfraorbitals) are small elements, narrow buffers between the orbit and the supramaxilla. Anterior to the orbit a large and outgoing lacrimal is paired with a smaller and more retiring lateral ethmoid. The latter is not a dermal bone, but is derived from the braincase, as the name suggests. There are no supraorbital bones in Amia, nor is there a jugal or quadratojugal. The sclerotic ring is also absent, although it is present in most other halecomorphs.
Behind and below the postinfraorbitals, Amia has no dermal covering, and some of the interior bones are revealed: the metapterygoid of the palate, and the quadrate and hyomandibular. Posterior to the quadrate, and parallel with it, is the symplectic. Both the quadrate and the symplectic articulate with the articular of the lower jaw. In fact, in Amia, the articular has two separate facets to receive these bones. How this works is unclear. The symplectic articulates with the hyomandibular which, in turn, is hooked into the opercular. Possibly, there are slightly different jaw movements associated with feeding and respiration, or the symplectic articulation is passive, helping to open the opercular gill cover when the mouth is closed and closing the gill cover when the mouth is open. In any event, the whole system is tied together through both the hyomandibula, internally, and the preopercular, externally, since all three operculars (the opercular, and the sub- and interoperculars) are tied to the preopercular, which parallels the symplectic.
 |
 |
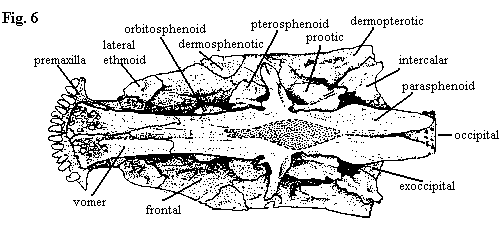 |
| Figures 4-6. Amia calva: skull in occipital view, braincase in left lateral and palatal views, with most dermal bones removed. See also, figure 8 showing dermal bones of the palate. Adapted from Grande & Bemis (1998). |
The occiput and posterior skull of Amia (fig. 4) suggest the shock troops of an invading army of dull-eyed, zombie panda bears emerging from a wormhole in space-time. Then again, this apparent likeness may merely reflect some personal psychosis of the author. The reader is invited to substitute an hallucination of his or her own choice. However, it must be something equally capable of sustaining the necessary minimum of brain function during the perilous, potentially lethal, passage through the tedium of occipital osteology.
The ears of the ursine storm trooper are formed by the posttemporals, which are mounted on the dermopterotics and the "parietals." The posttemporals do not have a tight articulation with the epioccipitals, which are tightly sutured to the parietals. The exoccipitals and the intercalars form the functional equivalent of a paroccipital process. The intercalars are tightly sutured to both the ventral processes of the posttemporals and to the exoccipitals, which are in turn firmly welded to the basioccipital. The intercalars are peculiar dermal bones of uncertain homology which are found only in Amia and teleosts among living fishes.
The occipital connection itself is formed by serial fusion of several vertebrae to form the occipital during development. The basioccipital forms the ventral margin of the foramen magnum, but it is hidden behind the occipital in the occipital view shown at right. In adults of Amiidae, the ventral processes of the exoccipitals do not reach the posterior end of the occiput because of the addition of vertebrae posteriorly (up to three, or even four, total centra in large individuals) to form the occipital. Amia lacks a pterotic, opisthotic (except as it may be related to the intercalar), supraotic, or supraoccipital.
The buccal cavity is lined with by gill arches, which bear numerous tooth patches. The arches are mounted on the enlarged ceratohyal, which also serves as the origin of the numerous branchiostegal rays. The lateral walls of the posterior skull are formed by the bones of the opercular series, as shown above.
The otic region of the braincase (fig. 5) is dominated by the prootic, and most of the other otic ossifications are lost. The prootics meet medially and are extensively sutured ventrally. However a large space on the ventral position of the otic capsule is open above the parasphenoid, and this forms part of the posterior myodome for the posterior or external rectus muscles of the eye. This myodome is absent in lepisosteiforms, but is further elaborated in the teleosts.
As in most actinopterygians, the parasphenoid (fig. 6) is the most prominent bone associated with the braincase and palate. In Amia, the parasphenoid runs from the occipital to a position below the anterior margin of the frontals, where it sutures with paired vomers (teleosts have a single, median vomer). About midway along the shaft of the parasphenoid, it produces dorsolateral ascending processes which attach to the ventrolateral edges of the sphenotics. A short, auxiliary process diverges from base of each ascending process and connects with the pterosphenoid anterolateral to the main shaft of the parasphenoid.
Anterior to the ascending processes of the parasphenoid are the pterosphenoids and the circular orbitosphenoids, which rest on the parasphenoid shaft. The ptero- and orbitosphenoids may be joined in larger animals. In most cases, two small basisphenoids (not shown in the figures) are found on the dorsal surface of the parasphenoid. These apparently mark an important articulation between the parasphenoid and the floor of the cartilaginous braincase itself. Lateral to the most anterior portion of the parasphenoid, the lateral ethmoids are attached to the anterolateral margins of the frontals. The lateral ethmoids, in turn, fix the position of the autopalatines (not shown) on the surface of the palate. At the anterior end of the palate, between the vomers and the premaxillae, are the pre-ethmoids, each of which forms a socket for an anterior articular process of the maxilla. Similar to the vomers, these pre-ethmoids become a single, median ventral ethmoid bone in teleosts.
 |
 |
 |
 |
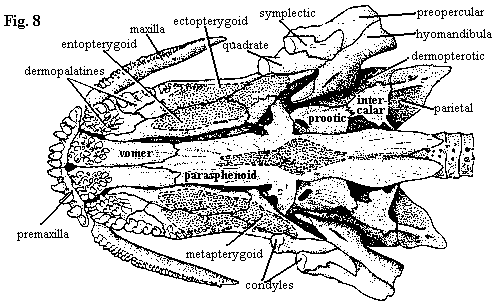
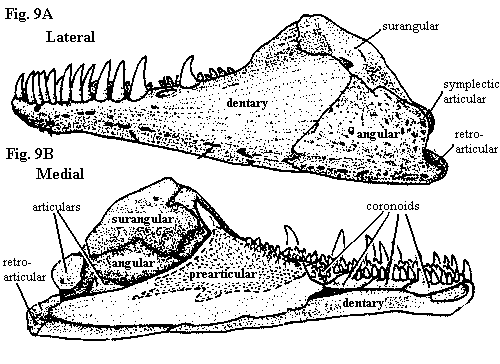
| Figures (7) Amia calva: lower jaw and branchiostegal apparatus in ventral view; (8) palate with dermal bones; (9) lower jaw in lateral and medial views; and (10) jaw and opercular apparatus in median view. Lower jaw and opercular elements are shown slightly disarticulated from upper jaw. All adapted from Grande & Bemis (1998). |
The branchiostegal rays (fig. 7) are asymmetrical, since one side must overlap the other below the jaw line. Both right- and left-handed individuals of Amia occur. There are 10 branchiostegal rays on each side. The last ray is referred to as the branchiopercular since, as discussed elsewhere, the branchiostegal rays essentially continue the opercular series onto the ventral surface of the lower jaw. The first two rays of the "dominant" side are generally fused. The gular occurs as a very large, unpaired, median plate.
The lower jaw (Figure 9) is complex and somewhat variable. The number of coronoids, for example, varies between 2 and 6 pairs. Amia also has the distinction of having no less than five separate ossifications arising from Meckel's cartilage: the two articulars, a retroarticular, coronomeckelian and mentomeckelian. The retroarticular is located in its usual spot at the posterior extremity of the jaw. It provides the attachment for large ligaments from the hyomandibular and interopercular. The corono- and mentomeckelian are located in the "pocket" formed by the prearticular and are not visible in fig. 9B. This pocket also encloses a space filled in life by the m. adductor mandibulae which attaches on the coronomeckelian. Since the coronomeckelian is so close to the quadrate articulation that it often fuses to the anterior articular, the mechanics of the arrangement are difficult to understand. Presumably, the symplectic articulation comes into play here. Note also that this is the reverse of the typical tetrapod condition in which the body of the muscle is located in or around the skull. Finally, the mentomeckelian is a tubular perichondral ossification at the anterior end of Meckel's cartilage.
The outer surface of the jaw (Fig. 9A) is formed by the dentary, angular and surangular G&B call it the supraangular). The dentary bears very large teeth. The mandibular symphysis is ligamentous, but tight, and is further reinforced by a tight fibrous connection with the massive gular plate. Very little motion is thus possible between the two dental rami.
Unfortunately, there is no single image which can adequately illustrate the way the entire jaw apparatus fits together. We have, in any case, just about run out of room to attempt it. Nevertheless, we will slip in Figure 10 which tries to combine enough information to convey the general idea. Note the rather tight connection between the opercular bones and the bones of the hyoid arch, forcing a coordination between jaw and opercular movements. In addition, the branchiopercular is the origin of the adductor mandibulae which inserts directly on the lower jaw. The net effect may be to force the operculum open when the jaw is closed and vice-versa as required to operate the "double pump" respiratory system. However, the mechanism must surely be more complicated. First, Amia is not well-designed for suction. Just opening the mouth is not a guarantee of lots of fresh water unless the fish is moving. Second, Amia does very well in poorly oxygenated waters -- or even out of water for long periods -- by gulping air. Perhaps the tight linkage is also derived as a way to bring the branchial tooth plates (see Figure 4) into play by using motions of the lower jaw to drive "chewing" by the branchial tooth plates.
The dental histology of Amia is somewhat unusual in that the dentine components contain pockets of living cells. In other respects, the tooth structure is unremarkable. The typical tooth consists of a pulp cavity surrounded by a dentine shaft. The tooth is covered by an outer layer of enamel-like hypermineralized tissue, referred to by the usual variety of names (enamel, enameloid, collar ganoine, etc.), all of which lack adequate chemical or structural definitions.
Essentially every oral surface in Amia is covered with teeth, like a porcupine turned inside-out. For example, the ventral surface of the parasphenoid, variably bears an extremely fine shagreen of tiny, sharply conical denticles, giving the bone surface a sandpaper-like texture. Larger toothlets are found on the anterior of the vomer, the palatal bones. The most impressive dentition is found on the premaxilla, which -- most unusually for an actinopterygian -- seems to have been widened expressly to accommodate more and larger front teeth. All of the marginal teeth are long, conical, and slightly curved towards the interior of the mouth. However, the maxillary teeth are noticeably smaller than the other marginal teeth. The coronoid teeth are smaller and also blunter than the large teeth of the dentary.
The title is minor test to see if anyone is still awake. Grande & Bemis inform us that Rafinesque (1815) rejected the name Amia because he thought it too short. Accordingly, I offer this modest substitute. It may be Classical Greek for the fish who is a survivor from ancient times. However, it might also mean the one who survived the ancient fish, as in last week's tuna salad sandwich. The ancient Greeks were, of course, unfamiliar with the dangers of tuna salad, presumably preferring to subsist on Greek salads. Greek salads carry their own risks, but Salmonella is not among them. We may thus conclude that the former definition applies.
But surely the problem is not that the name is too short, but that the genus is too long. Amia has been with us for about 60 million years. For the last half of that span, it has been the only living halecomorph. Both the skull and post-cranial skeleton are rather different from any other living fishes. We can't really conclude that the design is inferior. There is nothing obviously deficient about Amia, and it remains a very successful, voracious predator in eastern North America. Amia's range seems to have gradually contracted, but it is in no immediate danger of extinction. It seems to be a trophic and environmental generalist, with adaptability to a broad range of environments. Yet, Amia has not diversified in the manner of the Cichlidae or other inhabitants of low-energy fresh water environments, despite having a disjunct geographical distribution (its range appears continuous, but the waters of the Mississippi drainage basin have no contact with the various eastern watersheds which Amia also inhabits).
Grande & Bemis give us few hints to this puzzle, preferring a strictly empirical approach. But this is the question to be addressed. Why has Amia survived and, conversely, why only Amia? ATW021201.
checked ATW030131