|
|
Bones: Braincase |
| The Vertebrates |
Basisphenoid |
Braincase: Basisphenoid
Status
Like the ethmoid, the basisphenoid is a big and complex topic, intimately connected with the brain and the total design of the skull. Accordingly, after a very brief overview, we will content ourselves for the moment with a more detailed consideration of one portion of the bone: the sella turcica.
Overview
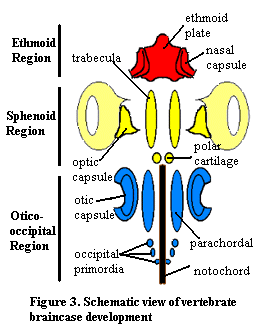
We may begin with what may now be familiar: an overview of the embryonic vertebrate braincase. The basisphenoid develops largely from the parachordals, mesodermal analogues of the trabecular cartilages, which are derived from embryonic neural crest tissue. Classically, it was supposed that the parachordals formed the basisphenoid, while the trabeculae formed the sphenethmoid and related bones. In fact, as we will see, the matter is not so simple.
The basisphenoid forms the floor of the braincase under the mid- and hind-brain in many vertebrates. It may extend forward under the forebrain as well. This is relatively common in tetrapods, since the anterior braincase of tetrapods is often unossified for reasons given elsewhere. In such organisms, the basisphenoid is the only ossified ventral element of the braincase. Under the braincase, the basisphenoid is integrated with the bones of the palate which, among many other functions, serve to support the basisphenoid on the marginal bones of the jaw, principally the maxillae. In turn, the basisphenoid acts as a strong longitiudinal beam for the palatal bones forming the roof of the mouth. See, for example the turtle skull at Skull of the Snapping Turtle.
In many cases, these anterior supporting roles are taken over by a dermal bone, the parasphenoid. In such cases, the basisphenoid plays a more posterior role as a broad plate, well integrated into the occiput and supporting the otic capsule. Another possible arrangement involving both bones can be seen in the plesiosaur figure at Plesiosaur Skull Anatomy. Frequently, the two are also indistinguishably fused (e.g. Thrinaxodon).
"Sella turcica" is Latin for "Turkish saddle." If this is an accurate description, we may marvel all the more at the success and longevity of the Ottoman Empire. The term is doubtless an early product of the same scientific whimsy that brought us Sonic Hedgehog, colored quarks, and Maxwell's Demon. Thus the sella turcica may never have seated Spahis (Ottoman -- and, later, French -- heavy cavalry). However, it does seat the adenohypophysis, also known as the anterior lobe of the pituitary. The sella is at a key point in the skull, as judged by several criteria. It lies almost at the geometrical center of the skull. It is just anterior to the rostral limit of the embryonic notochord, about at the junction of the trabeculae and parachordals, and also at the posterior limit of the forebrain. Dorsoventrally, it sits between the most ventral portion of the brain and the palate. The pituitary it protects is a union of brain tissue and pharyngeal ectoderm. Phylogenetically, the pituitary has deep connections with the nasal placodes and chemosensation. However, genetically, it is closer to the optic placodes and shares with them a number of gene regulation pathways, including pax6, six3, and lim3. Kondoh et al. 2000); Kioussi et al. 1999). In fact the adenohypophysis transiently expresses species- appropriate lens crystallins during development and can be genetically transdetermined to form a more or less normal lens. Kondoh et al. 2000). [2]
Developmentally, the sella turcica is a fairly simply cavitation in the surface of the basisphenoid. Apparantly, the posterior ends of the trabeculae are not actually parallel, as in the image above. Rather, they spread apart slightly so that there is a depression on the dorsal surface of the basisphenoid as it forms. I have not seen any reference to a similar separation of the parachordals, but it may turn out that this occurs. In any case, the result is that the basisphenoid has more than one embryonic origin. The anterior part is trabecular prechordal ectoderm and the posterior is neural crest parachordal tissue. Consequently, some writers refer to the bone as if it were two different ossifications: the "basipresphenoid" and "basipostsphenoid." The sella turcica itself is at the boundary between the two embryonic domains and includes contributions from both sources. Couly et al. (1993); Le Douarin et al. (1993). [3].
 All this antero-posterior patterning is interesting enough, but it is the dorsoventral interactions which have received all the press. This attention actually relates to the developmental molecular biology of the pituitary, rather than to osteology. In spite of having accumulated a stack of reprints and notes this high, your editor will be absolutely damned if he can make that subject particularly relevant to the present discussion. Furthermore, it amounts to little more than stamp-collecting at the moment because the system is far too complicated for our present, primitive grasp of bioinformatics and gene-level developmental regulation. In essence, the pituitary is the point at which all of the regulatory domains of the developing head overlap.
All this antero-posterior patterning is interesting enough, but it is the dorsoventral interactions which have received all the press. This attention actually relates to the developmental molecular biology of the pituitary, rather than to osteology. In spite of having accumulated a stack of reprints and notes this high, your editor will be absolutely damned if he can make that subject particularly relevant to the present discussion. Furthermore, it amounts to little more than stamp-collecting at the moment because the system is far too complicated for our present, primitive grasp of bioinformatics and gene-level developmental regulation. In essence, the pituitary is the point at which all of the regulatory domains of the developing head overlap.
For our purposes, a brief developmental overview is sufficient. See, generally, UNSW Embryo- Endocrine System- Pituitary. After neurulation is complete and the basic body plan establishes itself, the head structures begin to differentiate. The vertebrates are deuterostomes. That is, the gut is formed butt-first. The mouth and pharynx are derived by a secondary infolding of superficial ectoderm called the stomodeum. See Gilbert (2000: 490-492). The stomodeum is originally a simple layer of ectoderm blocking the anterior end of the gut. However, as the fore- and mid-brain differentiate from the anterior neural tube, they grow and inflect downwards. As a result, the ectodermal cells of the stomodeum are folded in under the brain and come close to the diencephalon. See Brain Function Diencephalon. A portion of the ectoderm associated with the roof of the stomodeum then invaginates dorsally, pinches off from the stomodeum, and migrates into physical contact with the base of the diencephalon at the site of the sella turcica. [4] This is Rathke's Pouch, which is the primordium of the adenohypophysis in most vertebrates. Eventually, the ectoderm of Rathke's Pouch differentiates into 5 to 7 different cell types which produce the various classes of pituitary hormones.
In almost all living vertebrates, all this activity has nothing at all to do with the development of the nasal passages. Sadly, our interest is mostly in dead vertebrates. So, we must digress from our discursion from our detour to touch on the nose. The nasal placodes are initially formed high up on the developing head. As the brain expands, they appear to migrate down and to the front of the face. At the same time, they begin to develop their own internal passages. These are lined by a thick layer of nasal epithelium surrounded by the mesenchyme (?) of the placode. In tetrapods, the orderly separation between nasal passages and pharynx breaks down quickly. The nasal and oral passages coalesce, and the adult nasal passage is formed by the construction of a secondary palate which repartitions the combined space. See, generally, Untitled Document.
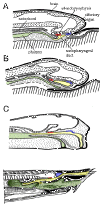
Now, finally, we have fashioned some crude conceptual tools with which to dissect the structure of earlier and more aberrant forms. The figure at right shows succesive stages in the development of the hagfish Myxine. These are adapted from Janvier (1986: 46-47), rescaled (and, in the case of 'C', redimensioned) for comparability. The hagfish has no forebrain inflection to speak of, and the conventional teaching is that the adenohypophysis develops from gut endoderm, so that the hagfish is unlike all other vertebrates. As the figure shows, this is may be incorrect. In fact, the nasopharyngeal duct -- an extension of the nasal cavity -- is in exactly the right place with what is probably ectodermal epithelium. Thus, we may suppose that the adenohypophysis has always been an ectodermal structure with the same basic genetic regulatory mechanisms in development. This supposition -- and the tight connection between the olfactory organs and the hypophysis -- is supported by the fact that both are derived from the anterior neural ridge, a very small area of ectoderm just in front of the anteriormost neural plate. Kouki et al. (2001); Takuma et al. 1998). Thus, what we are probably seeing in Myxine is not a nasopharyngeal duct, but a nasohypophyseal duct which happens to communicate with the pharynx. This sort of structure is quite common among early vertebrates.
There is no solid explanation for the function of this arrangement, but a glance at the known functions of the diencephalon (Brain Function Diencephalon) suggests many possibilities. The hagfish is guided largely by olfactory information. Hagfish have little ability to see or hear, and the hagfish brain primitive. It simply lacks the ability to integrate complex sense perceptions at a neural level. It is reasonable to suppose that much of the integration is therefore performed at a chemical level. The olfactory organ, connected with what passes for higher brain centers in the hagfish, might control immediate behavior: finding the food source or mate, fleeing the predator, etc. A downstream organ, part sensory and part endocrine, would receive direct sensory input from the environment and/or secondary messengers from the olfactory organ, and set in motion longer term preparations: to digest, to discharge gametes, to mobilize energy reserves or defensive mucous secretions. Similar arrangements, with or without connection to the pharynx, are found in the osteostracans, galeaspids, and probably heterostracans. The lamprey condition is essentially the same except for the peculiarity of the single median dorsal nostril.
In gnathostomes, the case is quite different. Here, the nasal placodes separate and move to opposite sides of the cranium. Presumably they are effectively pushed out of the way by the expanding brain. In any event, they lose contact with the hypophysis. So what happens to the old nasohypophysial duct? Why, nothing. It's still there. Recall that the very development which caused the stomodeum to fold inwards and separated the nasal placodes was the expansion of the forebrain. The logical consequence is that the median area of the anterior neural ridge is pushed right down into the mouth and becomes part of the infolding stomodeum. Presto! Instead of a nasohypophyseal duct invaginating from the face, we have Rathke's Pouch developing from the oral ectoderm. At the same time, and by the very same token, the enlarged forebrain permits increased neuronal procesing of olfactory information, so the tight chemical coupling between olfaction and endocrine release is no longer so necessary. The nasohypophyseal duct may thus be abridged and simplified. However, it is still apparently possible, albeit under rather bizarre experimental conditions, to induce chick embryos to form an ectodermal hypophyseal tube developing from the "face" even in the absence of a gut, much less a stomodeum. Withington et. al. (2001). The authors of that study conclude that the stomodeal ectoderm is induced by some other endoderm. It seems much more likely that the stomodeum has nothing to do with the case. Since brain inflexion is very limited in the absence of gut endoderm (see Id.) the old nasohypophyseal duct is simply making its way to the diencephalon from the outside, just as it did 400 million years ago.
[2] This is not one of those nearly useless experiments in which some tissue extract of unknown properties is used to create weird effects in ectoderm generally. See, e.g. Gleiberman et al. (1999). Kondoh used homozygous yot mutant zebrafish. These mutants have an altered gli2 gene which seems to lack specificity and shuts down several promoters in response to shh (Sonic Hedgehog). The most likely conclusion is that, up to the point of gli2 protein expression, presumptive lens and presumptive adenohypophysis are very similar, suggesting that they may be homologous if one goes back far enough. This is a particularly interesting speculation if one recalls that hagfish lack both lens and the ectodermal component of the adenohypophysis. All other living vertebrates (probably!) have both except those that are secondarily lensless.
[3] These papers are both products of the famous chicken-quail chimera experiments from the laboratory of Prof. Nicole Le Douarin (Commandeur dans l'Ordre de la Légion d'Honneur, Grand Officier dans l'Ordre National du Mérite, etc., etc. You don't see many academic resumes like this one!). Her wonderful experimental system is described in a brief and fairly non-technical manner by Gilbert 2000: 13).
[4] Physical contact is required for differentiation. At least some of the critical factors are not diffusible substances. Gleiberman et al. (1999). This ought to set off alarms. The warning here is that we must be very cautious -- far more cautious than many published papers -- in assuming that DNA transcription, RNA translation and protein activity are all co-localized. RNA transport, for example, is a well-known developmental phenomenon. The fact that RNA is made in one location does not mean that it expressed as protein in the same cell.
ATW 010804.
checked 020201
 All this antero-posterior patterning is interesting enough, but it is the dorsoventral interactions which have received all the press. This attention actually relates to the developmental molecular biology of the pituitary, rather than to osteology. In spite of having accumulated a stack of reprints and notes this high, your editor will be absolutely damned if he can make that subject particularly relevant to the present discussion. Furthermore, it amounts to little more than stamp-collecting at the moment because the system is far too complicated for our present, primitive grasp of bioinformatics and gene-level developmental regulation. In essence, the pituitary is the point at which all of the regulatory domains of the developing head overlap.
All this antero-posterior patterning is interesting enough, but it is the dorsoventral interactions which have received all the press. This attention actually relates to the developmental molecular biology of the pituitary, rather than to osteology. In spite of having accumulated a stack of reprints and notes this high, your editor will be absolutely damned if he can make that subject particularly relevant to the present discussion. Furthermore, it amounts to little more than stamp-collecting at the moment because the system is far too complicated for our present, primitive grasp of bioinformatics and gene-level developmental regulation. In essence, the pituitary is the point at which all of the regulatory domains of the developing head overlap.