Edaphosauridae
Taxa on This Page
- Edaphosauridae X
- Ianthasaurus X
The Edaphosauridae
Sphenacodont Cousins
An important evolutionary line of early synapsids springing from an ophiacodontid-varanopseid ancestry were the edaphosaurids. Advanced edaphosaurids were ecologically quite different from their contemporaries, the sphenacodontids. Unlike most basal synapsids, some edaphosaurids were herbivores, as is shown by the structure of the skull and teeth. In fact, they were perhaps the first tetrapod animals – certainly the first amniotes --to adopt a herbivorous lifestyle.
 Like many of their carnivorous sphenacodontid cousins, edaphosaurs were often equipped with a large “sail” along the back, formed by a great elongation of the neural spines of the vertebrae. This striking features, exhibited by several early synapsid lineages, is often said to have functioned as a temperature control device and perhaps also for sexual display.
See discussion below.
Like many of their carnivorous sphenacodontid cousins, edaphosaurs were often equipped with a large “sail” along the back, formed by a great elongation of the neural spines of the vertebrae. This striking features, exhibited by several early synapsid lineages, is often said to have functioned as a temperature control device and perhaps also for sexual display.
See discussion below.
Edaphosaurus, Ianthasaurus, and probably Glaucosaurus, can be placed with confidence in this family, but a number of lesser-known forms, like Lupeosaurus and Nitosaurus, may belong here as well. Ianthasaurus, Lupeosaurus, and Edaphosaurus are characterized by the presence of greatly elongated neural spines that are rounded in cross-section. Glaucosaurus is known only from a partial skull. Although it seems probable that Glaucosaurus had spines like those of Edaphosaurus, we have no proof as yet. The neural spines of Edaphosaurus and Ianthasaurus (but not Lupeosaurus) also bear well-developed lateral tubercles or crossbars. On the lower (proximal) part of the spine looks like the mast of a square-rigged sailing ship. Distally, the tubercles on the two sides become less regular and go out of register, so that the right and left tubercles emerge at different vertical levels. The tubercles also become shorter with increasing distance up the "mast". The arrangement of these tubercles along the height of the spines is similar in the two taxa, as is the loss of contact between the postorbital and supratemporal bones of the skull.
Despite their differences, edaphosaurids and sphenacodonts share a large number of cranial features, and are believed to be sister groups. The shared, derived characters of the two groups include the following:
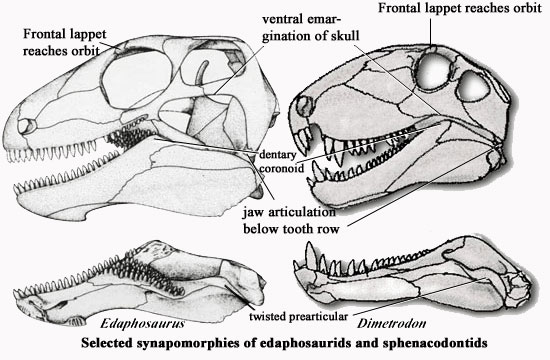
A lateral projection (lappet) of the frontal usually reaching the orbit;
-
ventral border of skull emarginated [H91];
-
A small quadratojugal which does not participate in the subtemporal bar [H91];
-
Jaw articulation below the tooth row [H91];
-
Rear of dentary with well-developed coronoid eminence [H91];
-
a prearticular twisted posteriorly as to underlie the pterygoideus process of the articular [MR90] [H91];
-
a pterygoideus process formed by the articular alone [MR90]; and
-
excavation of the lateral surfaces of the neural arches [MR90].
Within the edaphosaurids, Ianthasaurus is notably similar to sphenacodontids. For example, the lateral lappets of the frontal are short, but contact the orbit broadly. In Edaphosaurus, the opposite is true: the lateral tab of the frontal is long and contacts the orbit quite narrowly. The anterior process of the frontal is broad. The sides of the parietals are convex, as in Haptodus, rather than embayed, as in Edaphosaurus. The posterior processes of the parietals are directed straight back to form a median wedge, rather than angling laterally, as they do in Edaphosaurus [MR90].
 Ianthasaurus was almost certainly an insectivore. In any case it does not seem adapted to live on plants, and its short, blunt conical teeth are typical of bug eaters. As with so many groups of tetrapods of all kinds, the generalized small insectivorous form seems to have been the foundation on which selection built larger and more specialized animals [MR90]. The older literature often speaks of edaphosaurids evolving in the direction of herbivory from insectivory. However, it is unclear whether any of the edaphosaurid genera except Edaphosaurus ate their vegetables. Plant-eating may have been a late development in this lineage. Lupeosaurus doesn't seem to be built with the long, fat body of an herbivore -- but we lack the skull. Glaucosaurus doesn't have quite the dental specializations we might expect for eating plants -- but we lack the body. MAK, ATW081110
Ianthasaurus was almost certainly an insectivore. In any case it does not seem adapted to live on plants, and its short, blunt conical teeth are typical of bug eaters. As with so many groups of tetrapods of all kinds, the generalized small insectivorous form seems to have been the foundation on which selection built larger and more specialized animals [MR90]. The older literature often speaks of edaphosaurids evolving in the direction of herbivory from insectivory. However, it is unclear whether any of the edaphosaurid genera except Edaphosaurus ate their vegetables. Plant-eating may have been a late development in this lineage. Lupeosaurus doesn't seem to be built with the long, fat body of an herbivore -- but we lack the skull. Glaucosaurus doesn't have quite the dental specializations we might expect for eating plants -- but we lack the body. MAK, ATW081110
Edaphosauridae: Cope, 1882. Type: Edaphosaurus Cope, 1882. Other genera: Glaucosaurus Williston, 1915; Ianthasaurus Reisz & Berman, 1985 [MR90]; Lupeosaurus Romer, 1936 [S89]. Edaphosauridae incertae sedis include Edaphosaurus credneri Jaekel, 1910.
Range: Pennsylvanian to Cisuralian of Central and eastern U.S., central Europe (Germany).
 Phylogeny: Eupelycosauria ::: Sphenacodontia + * : Ianthasaurus + (Lupeosaurus + Glaucosaurus + Edaphosaurus).
Phylogeny: Eupelycosauria ::: Sphenacodontia + * : Ianthasaurus + (Lupeosaurus + Glaucosaurus + Edaphosaurus).
Characters:[1]
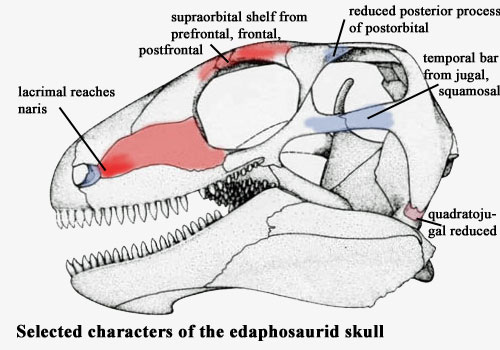
Rostrum: antorbital region tall [M94]; lacrimal reaches naris [RP40].
Skull table: skull table narrow [RB86].
Lateral skull: massive supraorbital shelf (bony “eyebrow”) [RB86]; posterior process of postorbital reduced, with loss of contact between postorbital and supratemporal; temporal bar from jugal & squamosal; skull margin emarginated under temporal fenestra [M94]; anterior process of quadratojugal reduced;
Occiput: supraoccipital smaller than in sphenacodonts [RP40].
Braincase: anterolateral wall of otic capsule formed by prootics [M95];
Mandible: “reflected” lamina of angular (characteristic of sphenacodonts and therapsids) not present;
Axial: body barrel-shaped; “sails” present [MR90]; only cervical centra have ventral keels [S89]; presacrals with elongated neural spines, often bearing multiple short transverse tubercles [S89] [MR90]; distal neural spines lack double-cylinder structure in cross section [HRS07]; neural spines circular in cross section distal to laterally compressed proximal section [BR86] [MR90]; neural spines with thickened cortex and central cavity [HRS07]; cervical neural spines lean  anteriorly & posterior spines lean posteriorly [RB86] [S89]; [MR90]; neural spines supported presumed "sails" (independently derived from sphenacodonts?); neural spine tubercles paired proximally (= "crossbars") [MR90];
anteriorly & posterior spines lean posteriorly [RB86] [S89]; [MR90]; neural spines supported presumed "sails" (independently derived from sphenacodonts?); neural spine tubercles paired proximally (= "crossbars") [MR90];
Appendicular: limbs short; clavicle expanded ventrally [S89].
Notes:
[1]We have tried to be cautious in using characters from Romer & Price [RP40]. Their “Edaphosauria” is a polyphyletic combination of caseids and Edaphosaurus. Consequently, their diagnosis of the group is a concatenation of plesiomorphic synapsid characters and highly specialized (but homoplastic) adaptations for eating plants. By contrast, their “Edaphosauridae” is limited to the genus Edaphosaurus. However, even with these severe limitations, [RP40] still contains a vast amount of useful information.
Image credits: The edaphosaurid skull is from [M95], the Dimetrodon skull from [H91].
 Ianthasaurus: Reisz & Berman, 1986. Type: I. hardestii [RB86].
Ianthasaurus: Reisz & Berman, 1986. Type: I. hardestii [RB86].
Range: Late Pennsylvanian [MR90] of Central U.S. (Kansas) [MR90].
Phylogeny: Edaphosauridae : (Lupeosaurus + (Glaucosaurus + Edaphosaurus)) + *.
Introduction: Ianthasaurus is a small edaphosaur from the Late Pennsylvanian that lacks many of the spectacular specializations seen in Edaphosaurus. Put another way, the characters that separate Ianthasaurus from Edaphosaurus are mostly adaptations for living on plants. [MR90]. For example, the marginal dentition of Ianthasaurus is similar to that of insectivorous reptiles, with slender conical teeth which are slightly recurved at the tips [BR86], and there is a slight development of a caniniform region. The palatal and mandibular dentition is unspecialized, and there are no batteries of teeth for crushing of plant materials. Also unlike Edaphosaurus, Ianthasaurus was lightly built and was probably quite agile. Ianthasaurus also shows many similarities in its cranial morphology to the small, primitive sphenacodont Haptodus. MAK, ATW081017
Characters: skull large [MR90]; skull long [RB86] [MR90][2].
Rostral skull: nasals long, trapezoidal, covering snout in dorsal view [MR90][5]; nasals with small anterior median process [MR90]; nasals reach nares [MR90]; maxilla long & low [MR90] maxilla participates slightly in naris [RB86]; lacrimal broad, reaching naris [RB86]; lacrimal posterior process stout, forming lower rim of orbit [RB86]; lacrimal posterior process with strong medially-directed shelf resting on tooth-bearing shelf of maxilla [RB86].
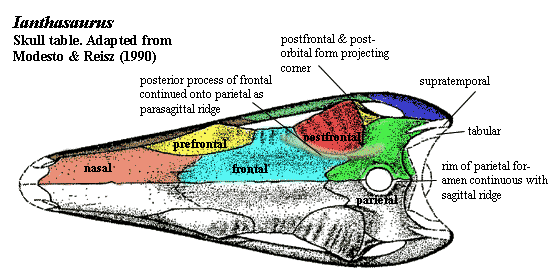 Skull Table: prefrontal triangular, with two sides forming orbital margin and skull table margin, 3rd overlapped by lacrimal [RB86][10]; nasal-frontal suture strongly interdigitating, overlain by anterior process of frontal [MR90]; frontal, prefrontal and postfrontal forming strong ridge above orbit, with all bones scalloped transversely (see figure) [MR90]; prefrontal with strong posterodorsal process participating in supraorbital ridge [MR90]; frontal long and broad anteriorly [MR90]; frontal lateral lappet strongly developed, located posterior to midpoint [MR90]; frontals with posterior processes participating in parasagittal ridges [MR90]; frontal-parietal suture interdigitating, overlain by small anterior process of parietal [MR90]; postfrontal expanded posteriorly, joining with postorbital to form distinct projecting “corner” of supraorbital ridge [MR90] [6]; parietal 2/3ds length of frontal [MR90]; parietal broadest at circular, rimmed parietal foramen [MR90]; lip on pineal foramen continuous with a short, median sagittal ridge perhaps for attachment of anterior sail [MR90]; parietal with ridge for supratemporal and groove for tabular [MR90]; small median postparietal present [MR90].
Skull Table: prefrontal triangular, with two sides forming orbital margin and skull table margin, 3rd overlapped by lacrimal [RB86][10]; nasal-frontal suture strongly interdigitating, overlain by anterior process of frontal [MR90]; frontal, prefrontal and postfrontal forming strong ridge above orbit, with all bones scalloped transversely (see figure) [MR90]; prefrontal with strong posterodorsal process participating in supraorbital ridge [MR90]; frontal long and broad anteriorly [MR90]; frontal lateral lappet strongly developed, located posterior to midpoint [MR90]; frontals with posterior processes participating in parasagittal ridges [MR90]; frontal-parietal suture interdigitating, overlain by small anterior process of parietal [MR90]; postfrontal expanded posteriorly, joining with postorbital to form distinct projecting “corner” of supraorbital ridge [MR90] [6]; parietal 2/3ds length of frontal [MR90]; parietal broadest at circular, rimmed parietal foramen [MR90]; lip on pineal foramen continuous with a short, median sagittal ridge perhaps for attachment of anterior sail [MR90]; parietal with ridge for supratemporal and groove for tabular [MR90]; small median postparietal present [MR90].
lateral skull: dorsal ramus of jugal thickened & continuous with orbital rim of postorbital [MR90]; jugal anterior ramus meeting posterior process of lacrimal on orbital rim [RB86]; jugal posterior process meeting squamosal to form temporal bar [BR86]; postorbital dorsal process short, not participating in dorsal orbit [RB86]; postorbital with small posterior process joining with parietal to form short posterior continuation of supraorbital ridge [MR90][6]; postorbital-supratemporal contact absent [MR90]; postorbital ventral process meeting jugal to form thin postorbital bar [RB86]; jugal participating broadly in ventral skull margin [BR86]; squamosal formed much (~50%) of oblong temporal fenestra [RB86]; temporal fenestra small [MR90]; skull margin concave under temporal fenestra [RB86]; posterior flange of squamosal supports supratemporal [MR90]; quadratojugal small, not participating in temporal bar [MR90]; jaw articulation at about same level as tooth row [RB86].
lower jaw: angular long, moderately deep, with ventral edge straight except gentle upward curvature in posterior 20% [MR90] [7]; anterior of angular divided into dorsal & ventral portions by prominent ridge [MR90]; angular not deep [RB86]; angular notch/flange absent [RB86]; coronoid process low [RB86]; coronoid bones absent [RB86]; prearticular, posterior end underlying pterygoideus process of articular [MR90] [9].
 Dentition: small caniniform teeth present [RB86] [MR90] [8]; maxilla bearing 27-29 sharply pointed, slightly recurved teeth [BR86] [MR90]; tooth plates absent [3] [MR90]; upper and lower teeth of same size [RB86].
Dentition: small caniniform teeth present [RB86] [MR90] [8]; maxilla bearing 27-29 sharply pointed, slightly recurved teeth [BR86] [MR90]; tooth plates absent [3] [MR90]; upper and lower teeth of same size [RB86].
Axial: body small [MR90]; trunk not expanded laterally [MR90]; 29+ presacrals [RB86$] [MR90]; “sail” relatively small [MR90]; presacral neural arches with lateral excavation [RB86] [MR90]; transverse processes short [RB86]; neural spines laterally compressed below 1st tubercles [RB86]; maximum 5 tubercles on each side of neural spines [4] [RB86] [MR90]; neural spines lack longitudinal ridges distal to 1st tubercles [MR90a]; neural spine tubercles angle slightly upward [MR90]; some proximal tubercles supported ventrally by slight webbing [MR90]; some proximal tubercles bearing accessory tubercles [MR90]; elongate cross-barred dorsal process on neural spine of axis [MR90$]; most anterior spines anteroposteriorly extended [RB86]; most anterior spines bent anteriorly, most posterior bent posteriorly [RB86]; tubercles on spines of anterior 2/3ds of presacral vertebrae only [RB86] [MR90]; dorsal centra slightly shorter than cervicals [RB86]; trunk ribs sharply curved proximally and not expanded [RB86] [MR90]; anterior dorsal and cervical ribs holocephalic [RB86]; sacral ribs very broadly expanded distally [RB86]; probably only 2 sacral ribs [RB86]; caudal ribs with short head, short shaft, and abrupt posterior curve [RB86]; caudal ribs dichocephalic [RB86].
Appendicular: posterior coracoid absent [RB86]; scapular blade rectangular, with deep notch at base of anterior margin [RB86]; anterior coracoid with broadly expanded convex anteroventral margin [RB86]; anterior coracoid with central anteroposterior ridge [RB86]; humerus long & narrow, with weak entepicondyle and absent supinator process [RB86]; humerus not “twisted” [RB86]; ilium, posterior process well developed and blade-like [RB86] [MR90].
Other: insectivorous [MR90] [1]. ATW081110 except as noted.
Romer vs. Homer
 ... and when the child of morning, rosy-fingered Dawn, appeared they again set sail for the host of the Achaeans. Apollo sent them a fair wind, so they raised their mast and hoisted their white sails aloft. As the sail bellied with the wind the ship flew through the wine-dark seas, and the foam hissed against her bows as she sped onward. Iliad I.
... and when the child of morning, rosy-fingered Dawn, appeared they again set sail for the host of the Achaeans. Apollo sent them a fair wind, so they raised their mast and hoisted their white sails aloft. As the sail bellied with the wind the ship flew through the wine-dark seas, and the foam hissed against her bows as she sped onward. Iliad I.
Introduction With the Usual Pretentious Drivel: Romer & Price [RP40] may have been under the impression that Edaphosaurus meant "ship lizard." In fact, εδαφο̣ς (edaphos) means "base," "bottom," or "foundation" -- perhaps a reference to its presumed phylogenetic position. The Greek root for "ship" is ναος (naos), more or less as in Latin (navis) or English (nautical). Romer may have been referring to Naosaurus, a poorly-known form of dubious affinities. In either case, the turn of phrase is best forgotten. We have serious doubts about any pelycosaur with foaming prow parting the wine-dark seas, etc.
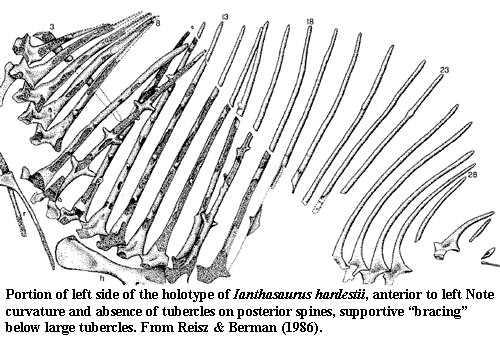
That said, there is no doubt that the cross-barred "sails" of some edaphosaurids are an interesting and unique feature. The sail is composed of greatly extended neural spines which were presumably joined by a relatively thick membrane. The reasons why we can presume this will become clear shortly. Many sphenacodonts, like Dimetrodon, had a similar structure. But the neural spines of Edaphosaurus bore tubercles sticking out at right angles. The tubercles nearest the base (or εδαφο̣ς if you must) came in pairs, one on either side of the neural spine, giving the appearance of cross-bars. However, further up on the neural spine, the tubercles of the left and right sides go increasingly out of register. In some of the largest species, the tubercles themselves bear tubercles, or may show traces of a sort of angle brace supporting the tubercle from below. By contrast, other edaphosaurids, such as Lupeosaurus, had no cross-bars of any kind.
Speculations, with More Drivel: Speculations about the purpose(s) of these structures are at least as varied as the sails themselves. Most of the proposed functions for edaphosaur sails sound suspiciously like the same reasons people historically go down to the sea in ships with sails: sex appeal, display, defense, transportation, and energy efficiency. The clear consensus today is in favor of energy efficiency, particularly thermoregulation, with sexual display favored as a secondary function. However, none of these hypotheses is completely consistent with the evidence, and most of the speculations come in at least two, inconsistent forms.

... and Finally, some Actual Data: First, here is what we think we know. In sphenacodontids, sail area scales with mass. In edaphosaurids, it doesn't. Sail area tends to get smaller relative to mass in the bigger species of Edaphosaurus [RP40]. However, relative sail area is smallest in the relatively small, generalized insectivore, Ianthasaurus [MR90]. In short, mass and sail area have no simple relationship in the Edaphosauridae. At the same time, the presence of a sail did mean a significant change in surface area. Hurlburt [H99] calculates that the presence of a sail increased the total body surface area by about 67% in E. boanerges. The linear dimensions of edaphosaur spines don't scale with anything. Spine length remains roughly constant throughout the clade [RP40].
While the sail may have had a radical effect on body area, it probably contributed very little to body mass. Hurlburt [H99] calculates that the sail included only 4% of body mass in E. boanerges. We are concerned that his assumptions are too conservative, but the figure is not likely to be all that much higher. There is not much evidence of extensive mechanical support, at least distal to the first crossbar.
At the same time, the sail cannot have been flapping around like pillow case on a laundry line. The evidence consistently points to a rather rigidly bound structure. Taphonomically, the spines are usually found parallel to each other, with extensive long-range order. That is, their order, front to back is often preserved. See, for example, the image of the fossil Ianthasaurus sail in the last section from [RB86]. That is not a reconstruction. That is the pattern on the slab as found. The other known specimen of this species had similar preservation [MR90]. Contrast this with the edaphosaurid skull, which is rarely found in articulation except for elements of the skull table; or the elements of the hand and foot which (so far as we are aware) have never yet been recovered in articulation in any edaphosaurid of any genus. Recovered spines are frequently broken, of course. However, surfaces showing breaks during life have not been noted -- with one interesting exception. Sumida [S89] notes the presence of a healed break in his specimen of Lupeosaurus. The implication, as he states, is that the spine was held rigidly enough for the broken ends to anneal.
A rigid material must be under tension, because it must resist gravitational forces. If the material is a vertical structure mounted on the back of a tetrapod, that tension will be significant. The sail must not only resist gravitational force, but the unpredictable forces generated by the movement of the animal. Tetrapods (particularly basal tetrapods) move forward by a process which can be modeled as rotation and counter-rotation about the animal's center of mass. The maximum forces on the sail will thus be at the front and back ends of the sail, and should scale as the moment of inertia. For animals of similar geometry, like edaphosaurs, the moment of inertia turns out to vary approximately as mass to the power 1.6 [WC02]. So, we'd expect to see signs of that strain increase dramatically as we go from Ianthasaurus to the largest known Edaphosaurus.
We do. Some of the traits we see are compensating adaptations: e.g., smaller heads, thicker spines, and a more even distribution of motive power between arms and legs. But other changes are just the obvious results of a lifetime of stress on bone [B97]. In the large edaphosaur E. pogonias, Romer & Price [RP40] note that the posterior spines are often bent so far that they actually point ventrally and "force their way downward into the area normally occupied by the short spines of the sacrals and proximate caudals” causing the latter to be abnormally formed, twisted or even split. They emphasize that this distortion was not pathological, since it is seen in every reasonably preserved specimen. However, this aberrant behavior is not seen in mid-dorsal spines of any species.
Structurally, edaphosaurid spines are rather different from those of Dimetrodon. Edaphosaurid spines have a thick cortex, a hollow core, and few vascular channels [H07]. This seems to be a typical biological design for strength and lightness, like the hollow bones of birds.
We should add that none of this explains anything about the crossbars or tubercles. Perhaps the tubercles of adjacent spines were connected by tissue. Perhaps not. There is simply no evidence.
But Enough Evidence. Back to Speculation: Given these more-or-less factual matters, how do the various speculations hold up? Not badly, on the whole -- except, oddly enough, some ideas about thermoregulation and sexual selection.
1) Physical protection against predation. Modesto & Reisz [MR90] offer this as one possible function of sails, adding that the largest tubercles would protect the vital epaxial musculature. The sails were not all that strong; but, then, neither were the predators. The jaws of Permo-Carboniferous carnivores look very different from those of Mesozoic and later predators. It seems very likely that they simply didn't work as efficiently. See our stunningly dull discussion of this topic at Varanopseidae (or, better yet, don't bother). If the sails were relatively thick and rigid, as we suppose, that may have been enough. This is particularly likely if the sails had evolved to withstand lateral forces. However ferocious they may have been, Permo-Carboniferous predators were not very tall.
2) Deterrence. The first cousin to the protection hypothesis is deterrence. Romer & Price [RP40] conceived this function in terms of intimidation. They were not persuaded, observing "the sail may have been of some slight protective value in that, in side view, the apparent bulk would be enormously increased, and thus perhaps a carnivore might be deterred from attacking. The authors cannot feel sure, however, that a sphenacodont would have been intelligent enough to have been deluded." It's a good point. We have mentioned that Early Permian predators were not all that strong. In addition, they were not all that bright. However, fraud has endless possibilities. The sail may have looked like a Permian palmetto plant. For that matter, perhaps the sail attracted, rather than repelled, attack. An indiscriminate attacker would then get nothing for its trouble but a mouthful of gristle laced wit h sharp bone fragments, while the edaphosaur would have little injured but its pride.
h sharp bone fragments, while the edaphosaur would have little injured but its pride.
3) Intraspecific Communication. Several ideas have been proposed based on this general theme, including individual recognition, sexual selection, intimidation of rivals. These kinds of specializations tend to be fairly species-specific. It would be a little odd -- but only a little -- if all members of this numerically important family developed this same specialization for intraspecific communications. Another discordant note is the absence (thus far) of any sign of sexual dimorphism. This doesn't get us very far. It simply underlines the point that no evidence whatsoever supports these speculations, or even suggests a way to test them.
4) Structural support. No reputable, current literature supports a structural role for the sail (although Romer is said to have favored this idea in the 1920's [B97]). Fortunately, we have no reputation to protect. The deafening silence on this point is based on the presumed insubstantiality of the sail membrane. As Bailey [B97] notes of sphenacodonts, “[t]he nondirectional stiffness imposed by the subcircular cross-section [in Dimetrodon] indicates that the spines were not subjected to the usual anteroposterior stresses of locomotion associated with anchoring points of muscles and ligaments.” But those parameters are only partially true in edaphosaurids. As mentioned, edaphosaurid spines are built differently, with a thick cortex, hollow core, and few vascular channels [H07] -- a formula for lightness and strength. They were certainly subject to major stresses toward the anterior and posterior ends. Furthermore, the proximal portions of the edaphosaurids spine are not "subcircular" in cross section, but show distinct lateral compression [MR90].
5)
Food Storage. This is another one from Romer & Price [RP40]. The biometric work of Bailey [B97] appears to eliminate this possibility. However, Romer & Price were not speaking of the whole sail, but only a layer between the epaxial musculature and the first cross-bar. Just keep this one in mind for a moment while we take on thermoregulation.
6) Thermoregulation. This the Current Best Guess. Many of the issues have been reviewed by Bennett [B96]. The theory comes in two basic flavors: radiative heating and convective cooling. The radiative heating flavor derives from work on sphenacodonts, which suggested that sails may have been able to speed up morning warming time by a substantial margin. However (a) the math works poorly for more realistic models; (b) heat transfer would be much slower, just when it is needed, because heat flow depends on blood flow (which is slower when the animal is cool); and (c) modern large lizards heat up relatively quickly without sails. In addition, the herbivorous Edaphosaurus had no obvious need for a quick morning warm-up.
The alternative is convective cooling: using bulk air movement to shed heat. Bennett [B97] tries to show how this works using a wind tunnel and a thin (3mm) aluminum plate welded to insulated pipe containing a heat source. Unfortunately, he measures only the
ΔT between arbitrary distal and proximal points on the plate, using constant power to heat the pipe. That only proves that a thin aluminum sheet makes a great radiator with forced convection and a large (unmeasured, but ~25 °K) ΔT between pipe and environment. What we suspect he ought to be measuring is how well the sheet cools the pipe, i.e. the power input needed to maintain a constant ΔT between pipe and environment. With that said, he does show that the aluminum does a much better job when it has "tubercles" -- even if the tubercles are made of wood. He suggests that the tubercles break up the boundary layer and make it turbulent, which sounds right. However we still don't know whether the stiff, relatively thick, and not particularly well-vascularized sail of edaphosaurids could actually shed heat generated in the body in the same way as a 3mm sheet of aluminum.
Conclusion: We don't really have a conclusion. The sexual selection and temperature regulation hypotheses are over-rated, but not necessarily wrong. As several writers have pointed out, the functions of the sail may have been different in different species, or served several functions at once.
Lacking a conclusion, we end with an Interesting Fact. The foundation of "warm-bloodedness" in mammals ("non-shivering thermogenesis") is the ability to burn fat quickly -- particularly a special type of "brown fat." The main depository of brown fat (particularly in small mammals and neonates) lies on the back, between the scapular blades. It is also the usual place for a hump in humped mammals. On an edaphosaurid, that's just under the point where the tubercles, and often the spines, are the most robust. Just as synapsids weren't all that bright, or strong-jawed, or agile, perhaps they had begun to develop non-shivering thermogenesis which wasn't very stable. If so, they would necessarily develop methods of shedding heat in parallel, and possibly in physical proximity, to the heat source. ATW081110.
Notes:
[1] based on dentition, large head, elongate body and small temporal openings [MR90].
[2] most comparative adjectives in the description refer for comparison to the better and earlier-known Edaphosaurus.
[3] The palate is poorly known, but tooth plates are clearly absent from the lower jaw.
[4] As in Edaphosaurus, the tubercles are not strictly paired and go increasingly out of register distally on the spine [MR90].
[5] A large, flattish nasal appears to be primitive for the clade Sphenacodontia + Edaphosauridae. It is reversed in Edaphosaurus and absent in Varanopseidae and in other more basal synapsids. We are not sure of the status in Ophiacodon.
[6] [MR90] note that [BR86] oriented the postfrontal backward, which had confused the restoration of this area.
[7] Dorsoventral striations on angular “suggest growth was occurring downwards, deepening the keel.” This is significant, since [MR90] note that both the holotype and the one significant referred specimen are probably juveniles.
[8] However, the figures in [MR90] show no obvious canine buttress on the maxilla.
[9] We note the following, so that no reader will have to repeat our wild goose chase on this topic. Modesto & Reisz [MR90] report that Reisz & Berman [RB86] found “a prearticular twisted posteriorly as to underlie the pterygoideus process of the articular.” In fact, Reisz & Berman reported “a prearticular twisted so as to underlie the pterygoideus process of the angular and [the] presence of a pterygoideus process formed by the angular.” This reference to the angular appears to be a replicated typographical error, since the pterygoideus (internal) process is correctly attributed to the articular elsewhere in Reisz & Berman’s paper. See also [H91] (same in Dimetrodon).
[10] The reconstruction by [MR90] is a bit different because the prefrontal is shown to be considerably bent to form a larger part of the strong awning of the skull table over the orbit.
 Like many of their carnivorous sphenacodontid cousins, edaphosaurs were often equipped with a large “sail” along the back, formed by a great elongation of the neural spines of the vertebrae. This striking features, exhibited by several early synapsid lineages, is often said to have functioned as a temperature control device and perhaps also for sexual display.
See discussion below.
Like many of their carnivorous sphenacodontid cousins, edaphosaurs were often equipped with a large “sail” along the back, formed by a great elongation of the neural spines of the vertebrae. This striking features, exhibited by several early synapsid lineages, is often said to have functioned as a temperature control device and perhaps also for sexual display.
See discussion below.
 Ianthasaurus was almost certainly an insectivore. In any case it does not seem adapted to live on plants, and its short, blunt conical teeth are typical of bug eaters. As with so many groups of tetrapods of all kinds, the generalized small insectivorous form seems to have been the foundation on which selection built larger and more specialized animals [MR90]. The older literature often speaks of edaphosaurids evolving in the direction of herbivory from insectivory. However, it is unclear whether any of the edaphosaurid genera except Edaphosaurus ate their vegetables. Plant-eating may have been a late development in this lineage. Lupeosaurus doesn't seem to be built with the long, fat body of an herbivore -- but we lack the skull. Glaucosaurus doesn't have quite the dental specializations we might expect for eating plants -- but we lack the body. MAK, ATW081110
Ianthasaurus was almost certainly an insectivore. In any case it does not seem adapted to live on plants, and its short, blunt conical teeth are typical of bug eaters. As with so many groups of tetrapods of all kinds, the generalized small insectivorous form seems to have been the foundation on which selection built larger and more specialized animals [MR90]. The older literature often speaks of edaphosaurids evolving in the direction of herbivory from insectivory. However, it is unclear whether any of the edaphosaurid genera except Edaphosaurus ate their vegetables. Plant-eating may have been a late development in this lineage. Lupeosaurus doesn't seem to be built with the long, fat body of an herbivore -- but we lack the skull. Glaucosaurus doesn't have quite the dental specializations we might expect for eating plants -- but we lack the body. MAK, ATW081110 Phylogeny:
Phylogeny:  anteriorly & posterior spines lean posteriorly [RB86] [S89]; [MR90]; neural spines supported presumed "sails" (independently derived from sphenacodonts?); neural spine tubercles paired proximally (= "crossbars") [MR90];
anteriorly & posterior spines lean posteriorly [RB86] [S89]; [MR90]; neural spines supported presumed "sails" (independently derived from sphenacodonts?); neural spine tubercles paired proximally (= "crossbars") [MR90];
 Skull Table: prefrontal triangular, with two sides forming orbital margin and skull table margin, 3rd overlapped by lacrimal [RB86]
Skull Table: prefrontal triangular, with two sides forming orbital margin and skull table margin, 3rd overlapped by lacrimal [RB86] Dentition: small caniniform teeth present [RB86] [MR90]
Dentition: small caniniform teeth present [RB86] [MR90]  ... and when the child of morning, rosy-fingered Dawn, appeared they again set sail for the host of the Achaeans. Apollo sent them a fair wind, so they raised their mast and hoisted their white sails aloft. As the sail bellied with the wind the ship flew through the wine-dark seas, and the foam hissed against her bows as she sped onward. Iliad I.
... and when the child of morning, rosy-fingered Dawn, appeared they again set sail for the host of the Achaeans. Apollo sent them a fair wind, so they raised their mast and hoisted their white sails aloft. As the sail bellied with the wind the ship flew through the wine-dark seas, and the foam hissed against her bows as she sped onward. Iliad I.
