Cetartiodactyla: Cetacea: Protocetidae
Taxa on This Page
- Protocetidae
Protocetidae: The Early Whales Turn Their Ankles
 There are, undoubtedly, many profound and wonderful things to be said about mammals in general and whales in particular. This essay will cover none of them. Instead, this will be one of those tiresome housekeeping sessions on the fine points of osteology. There is no help for it. Hans Thewissen and Philip Gingerich are excellent paleontologists who have recently changed our whole picture of cetacean evolution. Thewissen et al. (2001); Gingerich et al. (2001). Yet, in the process, they have tossed around so much terminology, mostly ankle terminology, that it is necessary to devote some space and time to a better understanding of the ankle of ferrungulates (= Ferae + Ungulata). Actually, it turns out that the ferrungulate tarsus is a fairly apt study for the understanding of ankles, assuming that one is apt to study such things at all. So, without further introduction or excuse we will begin.
There are, undoubtedly, many profound and wonderful things to be said about mammals in general and whales in particular. This essay will cover none of them. Instead, this will be one of those tiresome housekeeping sessions on the fine points of osteology. There is no help for it. Hans Thewissen and Philip Gingerich are excellent paleontologists who have recently changed our whole picture of cetacean evolution. Thewissen et al. (2001); Gingerich et al. (2001). Yet, in the process, they have tossed around so much terminology, mostly ankle terminology, that it is necessary to devote some space and time to a better understanding of the ankle of ferrungulates (= Ferae + Ungulata). Actually, it turns out that the ferrungulate tarsus is a fairly apt study for the understanding of ankles, assuming that one is apt to study such things at all. So, without further introduction or excuse we will begin.
The first difficulty is a pure question of nomenclature. Ankles are flexible and, like adolescents, adopt a number of incomprehensible attitudes depending on the pressures of the moment. How do we say what is dorsal, ventral, anterior, caudal and so on? Sadly, it turns out that different authors do it different ways, depending on their training, the organism under study, and the writer's usually misguided efforts to dumb things down. Here is the best we can do for now -- but don't try it on a pinniped, for example. Pick a spot towards the middle of the area that actually exerts force against the substrate, whether solid or liquid. Draw an imaginary straight line straight up the leg from that point. [2] Any viewpoint from which this line bisects the foot and/or ankle is plantar or ventral. Common, but wrong, synonyms are "posterior" and "caudal". Bear in mind that this rule applies even in the case of extreme digitigrade organisms in which a "ventral" or "plantar" view of the ankle in a natural life position requires one to look down on the tarsals and see the top side of the foot. The opposite side of the ankle and foot is always "dorsal." This terminology is actually quite logical because this surface is always dorsal during vertebrate limb development.
The trick is that this rule applies only up to the top of the tarsals. One centimeter more, and one passes out of the territory historically dominated by podiatrists and horse veterinarians -- over the border into the land of orthopods and locomotion specialists. These folk are oriented to the pelvis and to how all this connects to move the organism. Accordingly, their terminology is anteroposterior or caudo-cephalic. They look down (i.e., ventrally) on the horse and foot docs as old-fashioned quacks and like to borrow terms like revolute permitting rotation in one plane) from robotics and engineering.
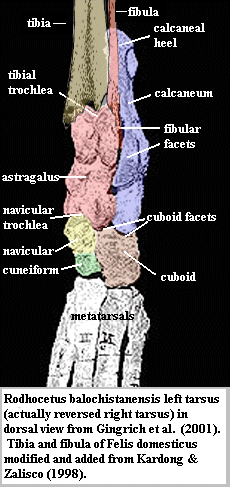
 Having adopted a suitable frame of reference, let us examine an actual ankle. The figure on the margin is the left ankle of the protocetid Rodhocetus balochistanensis Gingerich et al (2001) shown in dorsal view. [3] As a reminder, this means we are looking at the top of the foot and the front of the ankle bones as they probably occurred in life. We can start with the astragalus (red), sometimes referred to in mammals as the tibiale or talus. From this vantage, the astragalus is dominated by the tibial trochlea, a groove with curved sides at the top of the astragalus. The tibia has a process called -- with singular lack of imagination -- the dorsal projection, which fits in this groove. This is flanked by another process, the medial malleolus, which fits over the curve on the medial (left side of image) border of the trochlea. If we imagine the tibia as rigidly fixed, which it is not, this trochlea allows the foot to rotate up and down by at least 90° with respect to the tibia, and probably somewhat more. The navicular trochlea at the bottom of the astragalus extends that range by some further amount, and the cuneiform - metatarsal II articulation may extend the range a bit further yet. However, there is no obvious way for the foot to rotate left or right.
Having adopted a suitable frame of reference, let us examine an actual ankle. The figure on the margin is the left ankle of the protocetid Rodhocetus balochistanensis Gingerich et al (2001) shown in dorsal view. [3] As a reminder, this means we are looking at the top of the foot and the front of the ankle bones as they probably occurred in life. We can start with the astragalus (red), sometimes referred to in mammals as the tibiale or talus. From this vantage, the astragalus is dominated by the tibial trochlea, a groove with curved sides at the top of the astragalus. The tibia has a process called -- with singular lack of imagination -- the dorsal projection, which fits in this groove. This is flanked by another process, the medial malleolus, which fits over the curve on the medial (left side of image) border of the trochlea. If we imagine the tibia as rigidly fixed, which it is not, this trochlea allows the foot to rotate up and down by at least 90° with respect to the tibia, and probably somewhat more. The navicular trochlea at the bottom of the astragalus extends that range by some further amount, and the cuneiform - metatarsal II articulation may extend the range a bit further yet. However, there is no obvious way for the foot to rotate left or right.
So far, this looks like a 3-part hinge joint. But matters are not quite so simple. The lateral (facing away from the body) side of the ankle is occupied by the calcaneum, sometimes called the fibulare or calcaneus. For the moment, we will simply mark its salient features. The most obvious of these is the long calcaneal heel or calcaneal tuber, which projects proximo-ventrally from the proximal end of the calcaneum. At the opposite, distal end, the calcaneum articulates with the rather featureless cuboid, as does a corner of the astragalus. According to Gingerich et al., the cuboid has a concave, probably quite mobile contact with the astragalus. The articulation with the calcaneum, which we will see in more detail in a moment, consists of a marked dorsoventral notch -- shallow ventrally and very deep dorsally. It is not, however, a trochlea. The range of motion of the cuboid with respect to the calcaneum appears to be less than that provided by the navicular trochlea of the astragalus.
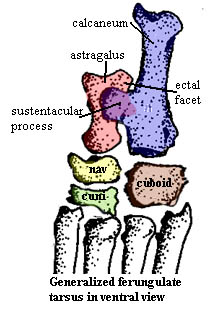
 Before looking at Rodhocetus in any more detail, we need to take a generalized peek at the ventral or plantar side of things. The figure on the left, of no particular animal [4], illustrates how the calcaneum projects a broad, medially directed process which supports the astragalus: the sustentaculum. The astragalus has a corresponding facet to receive the support, not surprisingly called the sustentacular facet. In addition, the enlarged lateral bulge of the tibial trochanter on the astragalus (the same that contacts the fibula) can also contact the calcaneum at an ectal facet.
Before looking at Rodhocetus in any more detail, we need to take a generalized peek at the ventral or plantar side of things. The figure on the left, of no particular animal [4], illustrates how the calcaneum projects a broad, medially directed process which supports the astragalus: the sustentaculum. The astragalus has a corresponding facet to receive the support, not surprisingly called the sustentacular facet. In addition, the enlarged lateral bulge of the tibial trochanter on the astragalus (the same that contacts the fibula) can also contact the calcaneum at an ectal facet.
With these basics out of the way, we can make more sense of the figure at right. This shows the astragalus and cuboid of Rodhocetus in dorsal, lateral and ventral views. The dorsal view is essentially the same as we saw above, except that the removal of the other bones allows us to see the details of the navicular trochlea and the deep cut in the cuboid which receives the calcaneum.
The lateral and ventral views show the astragalus from aspects normally covered by the calcaneum. Note the deep central groove which presumably accommodates, but does not bind, the body of the calcaneum. The fibula rides lightly on the outer surface of the tibial trochanter and is free to move between the tarsals as the tibia rotates on the trochanter. However, the calcaneum binds to the astragalus at the ectal and the elongate sustentacular facets, making it impossible for the two tarsals to slide past each other.
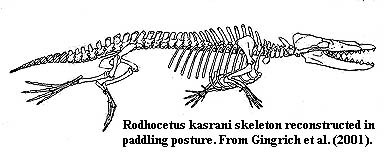 So how does it all work? We can see from the detail photographs that these bones are enormously complex works of geometry. The details are well beyond the scope of this essay. However, we can garner some basics from the figure at left. Notice how the calcaneal heel is drawn up parallel to the humerus during the course of the swimming power stroke. This is the work of the m. gastrocnemius which originates on the femur and inserts on the heel via the Achilles tendon. As the gastrocnemius contracts, it hauls up on the calcaneal heel, which distributes the force over the whole ankle via the broad sustentacular process, which, in turn, rotates the pes on the tibial trochanter of the astragalus.
So how does it all work? We can see from the detail photographs that these bones are enormously complex works of geometry. The details are well beyond the scope of this essay. However, we can garner some basics from the figure at left. Notice how the calcaneal heel is drawn up parallel to the humerus during the course of the swimming power stroke. This is the work of the m. gastrocnemius which originates on the femur and inserts on the heel via the Achilles tendon. As the gastrocnemius contracts, it hauls up on the calcaneal heel, which distributes the force over the whole ankle via the broad sustentacular process, which, in turn, rotates the pes on the tibial trochanter of the astragalus.
That much seems straightforward. But the fine points separate human engineering from the work of evolution. The groove in the cuboid which receives the calcaneum is not quite symmetrical, and the force applied through the sustentacular facet is a bit off center. As we have seen, the articulations of the astragalus with the distal tarsals also seem somewhat more flexible than those between the calcaneum and cuboid. These are small effects, but they add up to a slight tendency for the dorsal face of the astragalus to rotate medially under tension. This effect is magnified by the substantial length of the digits and the greater flexibility of the distal articulations of the astragalus. The resultant is seen in the right foot of the swimming image: digits II and III would be rotated up and behind digits IV and V, reducing the effects of water resistance during the recovery stroke. Note that water resistance would keep the foot rotated and the system under tension until the femur was retracted. At that point -- at the end of the recovery stroke -- water resistance would be reduced and the inner foot would tend to return to the same plane as digits IV & V just in time for the next power stroke. ATW 011017
Note: [2] You may draw a real line, if you insist, but the procedure is not recommended in the case of most carnivores, rabid hamsters, mad cows, etc.
[3] Actually this is a mirror image of the right ankle. The tibia and fibula were not illustrated. The tibia and fibula of a domestic cat have been faked in to show the general scheme. The original epipodial bones were undoubtedly a bit more robust.
[4] This illustration began life as a cat from Kardong & Zalisco (1998). However real cats have multiple cuneiforms, a fairly small sustentaculum, and other distractions. ATW030125.
 Making the Grade: a Digression on Darwin and Hennig
Making the Grade: a Digression on Darwin and Hennig
All discussions of whale phylogeny seem to proceed by phyletic grades. The reason for this is not (as we had first supposed) that whale workers are a reactionary lot who refuse to adopt a phylogenetic approach out of sheer, obstinate perversity. No, the problem is that most whales are very large, mobile and marine. As a result, populations tend to be genetically continuous. Every whale of a given species has a realistic chance of mating with nearly every other member of the species of suitable age, sex and disposition. In this kind of population, evolution is unlikely to proceed by random genetic drift in isolated populations. It is more likely to occur by Darwinian selection operating rather uniformly on the whole species. Thus we observe only a small number of genera, which follow broad patterns of change operating in a consistent manner over long periods of time.
Classical, descriptive paleontology is very good at dealing with this sort of pattern. Conversely, cladistics may fail because synapomorphies are few and homoplasy ("convergent evolution") is common [1]. This point is more subtle than it appears, and goes to the heart of the difference between classical Darwinian evolutionary theory and Hennigian cladistics. These are, of course, simply different aspects of the same core ideas. But Charles Darwin was a naturalist, who studied birds, as well as other organisms fairly high up on the Nineteenth Century Scala Naturae. Willi Hennig was an entomologist. He studied bugs. Darwin grew up with a firm understanding that great purposes, reasoned law, and orderly progress governed the British Empire and the universe at large. Hennig grew up in the nihilism and chaos of Germany between the World Wars. These divergent experiences may have left their mark on the way these scientists approached their common subject. We are fortunate that this is so.
The Hennigian paradigm is best suited for discussing populations which (a) are easily isolated and (b) are sensitive to microenvironmental change. In such cases, random genetic changes are quite likely to become fixed in a population, purely by chance. That is, one mutant variant of a gene (allele), having no selective advantage, may become the only variant in the population simply because the "normal" allele dies out by chance. Statistically, this kind of event is far more common than one might intuitively suppose. It is even more likely if the population (like many insects) is periodically subject to wide fluctuations in number, perhaps because of sensitivity to small environmental fluctuations. In the extreme case, where only one breeding pair of a population is left, any homozygous alleles the pair have in common will become fixed in one generation, to a mathematical certainty.
Sooner or later, this process will result in a population in Valley A that is no longer capable of mating with their cousins in Valley B. Even in the absence of functional change, each population has acquired a set of shared, derived characteristics (synapomorphies) which distinguishes it. However, an absolute functional neutrality is quite unlikely. In virtually every real case, the A bugs will be a bit better at chewing oak leaves, while B bugs thrive on ash; A bugs will survive better in dry seasons, while B bugs love rain; or A bugs will better avoid predation from birds, while B bugs better avoid frogs. Given these small differences, the bugs' ecospace will be partitioned if the populations are joined again. Note that this occurs even if Valleys A and B are identical. The basic divergence and fundamental synapomorphies were acquired purely by chance, and not by selection. Selection merely refines the population niches when they are later mixed (again by chance). Indeed, because the bugs are very sensitive to random changes in microenvironment, the population effects of these random fluctuations will effectively mask all but the most potent or most long-term selective pressures. When such pressures are felt, the A and B bugs have slightly different genetic starting points, by chance, and are likely to evolve along divergent paths in response to that selective pressure.
Contrast this case with finches or -- even better -- whales. Single, worldwide [2] populations are common. Because (until recently), whale populations were rather large, as well as global, both genetic isolation and random fixation of mutant alleles was rare. An individual whale's microenvironment tends to be very stable, since (a) it is averaged over vast areas and long lives and (b) the whale is fully capable of moving away from adverse local conditions. In this type of population, mutations will tend not to become fixed randomly because isolation and population fluctuations are rare. As a result, synapomorphic characters are few. Conversely, longer-term selective pressures will not be masked. We are quite likely to observe, and do observe, gradual adaptive changes over time without much speciation -- classic Darwinian selection.
To a limited extent we may also observe a converging effect between species. Again, since whales are large and mobile, members of different species will tend to experience the same macroenvironmental selective pressures in the same way. Because of the averaging effect of great space and long lives, the average environment experienced by individual members of different species may also converge. If so, selective pressures on the two species will tend to be similar in nature and extent. Further, large, stable populations are limited in their ability to generate genetic novelty precisely because random mutations are rarely fixed. Selection is more likely to proceed from small incremental changes on a relatively conservative body plan. Thus, even after two whale species have diverged, they will tend to experience similar selective pressures, and respond from a repertoire of genetic variation biased against qualitative alteration of the shared body plan. As a result, convergent adaptation is more likely than one might expect from chance alone.
The net result is that whales tend to be Darwinian creatures: not hostile to change, but conservative, conformist, orderly and immune to fashion in their response -- rather the image of the Nineteenth Century Briton in fact. Hennig's insects are more prone to randomness and extremes, to the best and worst of novelty and creativity -- perhaps more like interbellum Germans. [3] As matters turned out, Hennig's paradigm is probably much more often applicable than Darwin's. However, whales are Darwinists and are appropriately treated from that conceptual reference point. [4]. ATW030201.
Notes: [1] For one interesting example, see Uhen (1999). The classical story is that the protecetids were succeeded by the long-bodied Basilosauridae. One branch of this group, exemplified by the shorter-bodied Dorudon, gave rise to modern whales. For reasons we will get into when we discuss locomotion, this makes excellent sense. However, Uhen's cladistic analysis finds that Dorudon and Basilosaurus define a monophyletic group to the exclusion of modern whales. This clade seems to have no synapomorphies, and constitutes one of the few phylogenetic possibilities that we can almost certainly reject out of hand.
[2] Actually, hemisphere-wide. Many species have Northern and Southern hemisphere populations which do tend to diverge.
[3] These observation are, of course, made by one who grew up in the somewhat studied and self-conscious pluralism of Late Twentieth Century America. We are none of us immune from making the world in our own image.
[4] We should add an important qualification. In the Late Oligocene, the a fundamental split occurred between the Odontoceti and the Mysticeti. The former group also gave rise to various endemic lines of river dolphins. These developments created much greater opportunities for genetic isolation and meaningful partition of ecospace. Accordingly, the pattern from this point forward (generally for the Autoceta) is less Darwinian and more Hennigian.
In many ways, the cetacean ear is radically different from the ear of terrestrial mammals. A channel of fat and bone in the lower jaw replaces the external ears and auditory meatus of land dwellers. The relict external ear passage is squeezed shut and, in mysticetes, is further physically plugged with a "glove finger" of soft tissue. The whale has no tympanic membrane, and the ear is, in fact, carefully insulated from airborne sound by a grossly inflated auditory bulla. The middle ear ossicles are frequently attached, even fused, to the bulla or petrosal, rather than being lightly suspended in an epitympanic recess. Only the inner ear is relatively unchanged.
In spite of these extensive differences, the evolutionary pattern follows the Darwinian paradigm quite well. In the jaw, cetaceans begin with the standard Meckelian foramina. These expand and multiply.Standard connective tissues develop into specialized fat pads which conduct sound along the expanded channel. The shape and surface of the surrounding bone gradually develops to perform the role of acoustic insulation for high frequency sound passing to the ear.
The connection of this channel with Meckel's cartilage. Is hardly fortuitous. The posterior end of the cartilage ossifies as the ectotympanic, the bone forming the tympanic ring which holds the tympanic membrane. The posterior process of this same bone forms part of the bony ventral surface of the external auditory meatus. In pakicetids (see figure at right), the ectotympanic has already overgrown, giving rise to a large bulla surrounding the petrosal.
The auditory bulla is relatively standard issue for mammals. However, in whales, the bulla gradually becomes very large and dense (pachyosteosclerotic) for improved sound insulation. The structure of the cetacean bulla is unique. The series of adaptive changes involved are discussed in some depth by Luo (1998). Luo's review is highly recommended, and no attempt will be made to cover the details here. More derived products of this process may be seen in the Georgiacetus bulla figure and the mysticete bulla figure (also reproduced at Mysticeti). The external auditory meatus is squeezed shut between the conical apophysis and the sigmoid process. The tympanic ring itself gradually rotates away from its tetrapod functional orientation (as do the auditory ossicles). The tympanum itself becomes a ligament of uncertain but apparently important function, while the ossicles attach either to the petrosal or the bulla. The size and degree of enervation of the cochlea gradually increase.
These events all seem to happen in a regular, Darwinian way. There are few aberrations or exceptions. From the point of view of ear anatomy, we might ignore the species concept entirely, and treat cetacean ear evolution as a stepwise addition of adaptive changes on a single population throughout the long Eocene interval. ATW030201.
Descriptions
 Protocetidae: Artiocetus, Babiacetus, Eocetus, Gaviacetus, Indocetus [3], Natchitochia, Pappocetus, Protocetes, Rodhocetus, Takracetus. Used (somewhat idiosyncratically) here to mean all cetaceans which are primarily marine or, to give it a more formal definition, Rodhocetus + Physeter. This seems to be the definition applied by Uhen [U99] and others. However, the literature for this group contains no satisfactory explicit definition and often expressly
includes the pakicetids. Huber et al. [H+98] suggest that it means cetaceans which spent no significant time on land and lacked weight-bearing hind limbs. That may have been the consensus view, but now it turns out that Rodhocetus had very substantial hindlimbs -- perhaps a textbook case of why apomorphy-based definitions are unworkable.
Protocetidae: Artiocetus, Babiacetus, Eocetus, Gaviacetus, Indocetus [3], Natchitochia, Pappocetus, Protocetes, Rodhocetus, Takracetus. Used (somewhat idiosyncratically) here to mean all cetaceans which are primarily marine or, to give it a more formal definition, Rodhocetus + Physeter. This seems to be the definition applied by Uhen [U99] and others. However, the literature for this group contains no satisfactory explicit definition and often expressly
includes the pakicetids. Huber et al. [H+98] suggest that it means cetaceans which spent no significant time on land and lacked weight-bearing hind limbs. That may have been the consensus view, but now it turns out that Rodhocetus had very substantial hindlimbs -- perhaps a textbook case of why apomorphy-based definitions are unworkable.
from the Early Eocene [FB94], or perhaps middle Eocene [H+98], as Rodhocetus is from the early Middle Eocene (Lutetian). [G+94].
primitively less than 3 m; preorbital rostrum primitively <60% of skull length [H+98]; nares primitively only slightly retracted on rostrum [G98] [U98]; nares retracted at least to level of canines [G+94]; supraorbital process formed primitively by frontals [H+98] [T98]; primitively with laterally facing orbits covered by a supraorbital shield [T98]; isolation of petrosal limited (directional hearing questionable) [GU98]; large, dense auditory bullae [GU98]; bulla with median furrow on ventral surface [L98$]; bulla posterior process attached by double pedicels separated by a foramen (image) [L98$]; bulla posterior process is thick horizontal plate covering (and sutured to) entire posterior process of petrosal [L98$]; "incipient" conical apophysis [L98$]; bulla opening for external auditory meatus reduced [L98$]; sigmoid process pachyostosed, and twisted, with involuted margins [L98$]; incus mallear facet faces anterolaterally [TH93]; primitively lacking pterygoid fossa or accessory air sinuses [G+94]; EQ perhaps 0.25, compared to comparable terrestrial mammals [G98]; endocranial retia present? [T98]; mandibular symphysis long, terminating ventral to p2-3 [H+98]; dentary with series of mental foramina along buccal side [H+98]; dentary with enlarged mandibular foramen opening into deep posterolingual fossa (wave guide for underwater hearing) [G98] [H+98]; $? mandibular foramen much enlarged to transmit underwater sound to middle ear [BT98] [T+01] [G+94] [GU98]; "ascending ramus" (coronoid process of dentary, we suppose) concave medially [BT98$]; dentition reduced, but generally similar to mesonychids; functional occlusion and heavy wear retained (food was chewed) [GU98]; P1-2 with 2 roots & rest of cheek teeth with 3 (primitive number); P3-4 with 2 buccal cusps [H+98]; P3-4 with strong central paracone & small metacone [H+98]; P3 large, transversely wide distally & 3-rooted [H+98]; P4 larger than M1 in occlusal outline [$T98]; upper molars wide, 3-rooted [H+98]; upper molars with large parastyle [T98]; upper molars with low or vestigial protocones [H+98]; retention of M3 [T98]; lower cheek teeth with distinct talonid & trigonid, each bearing only 1 cusp [BT98]; metaconid absent [T98]; cervical centra shortened to 75% or less length of anterior thoracic centra [H+98] [G+94]; cervical centra somewhat shortened, with length less than diameter [G98]; zygapophyses steeply inclined toward the vertical [U98a]; distal part of prezygapophyses expanded [U98a]; primitively retained some remnants of central arch of thoracic vertebrae [G98]; thoracic vertebrae primitively with tall neural spines (forelimbs potentially weight-bearing) [G98]; thoracic vertebrae with rib tuberculum facet on transverse process & rib capitulum facet on anterolateral part of centrum [U98a]; capitular & tubercular facets well separated on anterior vertebrae, but posteriorly, transverse processes arise from centrum and facets merge [U98a]; lumbar spines with stout neural spines (tail musculature) [G+94]; lumbar transverse processes based on centrum, becoming more ventral in posterior vertebrae [U98a]; lumbar vertebrae may be somewhat elongated (Eocetus) [U99]; sacrum composed of less than four vertebrae fused to pelvis ["reduced" U98] [$T98] [1]; sacral vertebrae not fused to each other [G+94]; tail flukes present in later forms [GU98]; strongly developed deltopectoral crest [T+01]; retained substantial pelvic girdle [G98]; pubis may elongate relative to ilium & ischium [U99]; hind limbs primitively reduced but functional [GU98] [H+98]; femur short when present [H+98] [G+94]; femur primitively robust [G98]; femoral head primitively large & spherical, with fovea for ligamentum capitis femoris [G+94]; tarsus primitively as pakicetids [G+01]; some tendency to increase bone thickness and density [M98] [U99 (Eocetus as an example)]; caudal undulation for swimming in all [G+01]; found in deep neritic zone [G+94].
Notes: [1] There is considerable variability here. Rodhocetus, for example, has 4 easily identified sacral vertebrae. However, they are not fused, although they do show evidence of considerable contact through their transverse processes [U98]. [2] [G+94] is the Rodhocetus paper. Since we have anchored this taxon on Rodhocetus, some of the characters of this genus which are shared with later whales are listed here as characters of the clade. [3] Bajpai & Thewissen (1998) manage to suggest (without saying so) that Indocetus is a nomen dubium with everything referable to either Rodhocetus or Remingtonocetus. [4] First cetaceans to leave the eastern end of the Tethys Sea (India & Pakistan).
Links: Cetaceans; Origin of Whales.pdf; Wal und Mensch | Kommentar zu Schulze (1996) (German); ?????????? Chinese); The Emergence of Whales, Chp. 1; The Scientist :: Of hippopotami and whales; Rodhocetus; Untitled Hungarian); Molecular Evolution.ppt; Almost like a whale; WHALE FOSSILS AND EVOLUTION - Enchanted Learning Software; New Fossils Resolve Whale's Origin: Science News Online, Sept. 22, 2001; StudyWorks! Online : From Where the Whale?; Evolution of Whales @ nationalgeographic.com; Cetacea; Mark D. Uhen, CT Scans.
References: Bajpai & Thewissen (1998) [BT98]; Fordyce & Barnes (1994) [FB94]; Gingerich 1998) [G98]; Gingerich & Uhen (1998) [GU98]; Gingerich et al. (1994) [G+94]; Gingerich et al. (2001) [G+01]; Hulbert et al. (1998) [H+98]; Luo (1998) [L98]; Madar (1998) [M98]; Thewissen 1998) [T98]; Thewissen et al. (2001) [T+01]; Thewissen & Hussain (1993) [TH93]; Uhen 1998) [U98]; Uhen (1998a) [U98a]; Uhen (1999) [U99]. ATW030126.
 There are, undoubtedly, many profound and wonderful things to be said about mammals in general and whales in particular. This essay will cover none of them. Instead, this will be one of those tiresome housekeeping sessions on the fine points of osteology. There is no help for it. Hans Thewissen and Philip Gingerich are excellent paleontologists who have recently changed our whole picture of cetacean evolution. Thewissen et al. (2001); Gingerich et al. (2001). Yet, in the process, they have tossed around so much terminology, mostly ankle terminology, that it is necessary to devote some space and time to a better understanding of the ankle of ferrungulates (= Ferae + Ungulata). Actually, it turns out that the ferrungulate tarsus is a fairly apt study for the understanding of ankles, assuming that one is apt to study such things at all. So, without further introduction or excuse we will begin.
There are, undoubtedly, many profound and wonderful things to be said about mammals in general and whales in particular. This essay will cover none of them. Instead, this will be one of those tiresome housekeeping sessions on the fine points of osteology. There is no help for it. Hans Thewissen and Philip Gingerich are excellent paleontologists who have recently changed our whole picture of cetacean evolution. Thewissen et al. (2001); Gingerich et al. (2001). Yet, in the process, they have tossed around so much terminology, mostly ankle terminology, that it is necessary to devote some space and time to a better understanding of the ankle of ferrungulates (= Ferae + Ungulata). Actually, it turns out that the ferrungulate tarsus is a fairly apt study for the understanding of ankles, assuming that one is apt to study such things at all. So, without further introduction or excuse we will begin. Having adopted a suitable frame of reference, let us examine an actual ankle. The figure on the margin is the left ankle of the protocetid Rodhocetus balochistanensis Gingerich et al (2001) shown in dorsal view.
Having adopted a suitable frame of reference, let us examine an actual ankle. The figure on the margin is the left ankle of the protocetid Rodhocetus balochistanensis Gingerich et al (2001) shown in dorsal view. 
 So how does it all work? We can see from the detail photographs that these bones are enormously complex works of geometry. The details are well beyond the scope of this essay. However, we can garner some basics from the figure at left. Notice how the calcaneal heel is drawn up parallel to the humerus during the course of the swimming power stroke. This is the work of the m. gastrocnemius which originates on the femur and inserts on the heel via the Achilles tendon. As the gastrocnemius contracts, it hauls up on the calcaneal heel, which distributes the force over the whole ankle via the broad sustentacular process, which, in turn, rotates the pes on the tibial trochanter of the astragalus.
So how does it all work? We can see from the detail photographs that these bones are enormously complex works of geometry. The details are well beyond the scope of this essay. However, we can garner some basics from the figure at left. Notice how the calcaneal heel is drawn up parallel to the humerus during the course of the swimming power stroke. This is the work of the m. gastrocnemius which originates on the femur and inserts on the heel via the Achilles tendon. As the gastrocnemius contracts, it hauls up on the calcaneal heel, which distributes the force over the whole ankle via the broad sustentacular process, which, in turn, rotates the pes on the tibial trochanter of the astragalus. Making the Grade: a Digression on Darwin and Hennig
Making the Grade: a Digression on Darwin and Hennig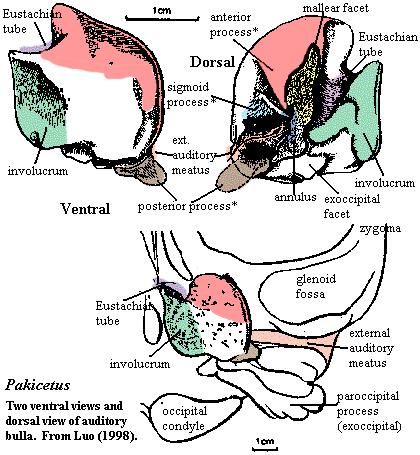 Water Music and the Darwinian Ear
Water Music and the Darwinian Ear