Abstract
This page describes the metazoan animal) evolutionary phenomenon popularly known as the "Cambrian explosion." The apparent discrepancy between divergence ages implied by genetic calibration techniques and a literal interpretation of the fossil record is discussed.
Keywords: Cambrian, Cambrian biota, Cambrian explosion, Cambrian radiation, fossil record, evolution, Metazoan radiation
Introduction
The abrupt appearance of a diverse and highly derived fauna in the brief Tommotian and Atdabanian Ages of the Terreneuvian and Cambrian Epoch 2 (respectively) is widely known as the 'Cambrian Explosion.' Although that particular phrase only came into common usage in the early to mid 1970s, the event itself has long been recognized as a phenomenon demanding some accommodation from evolutionary theory. As early as 1859, Charles Darwin drew attention to the matter in Origin of Species, and it is probable he had considered the matter for many years prior to that.
However, despite the rapid proliferation of evolutionary novelties which undoubtedly occurred at this time, at least some of the phenomenon is attributable to the acquisition of preservational characteristics - 'hard parts' - and multiple lines of evidence reveal that life was already highly diversified prior to the Tommotian. The fossil record is continually yielding more and better evidence of pre-Tommotian life, from the earliest biochemicals extracted from ~3,800 Ma cratonic rocks of southwest Greenland, through the perplexing grotesquery of the Ediacarans, to the strangely quiescent Fortunian age, when we find little more than a few small, undistinguished shelly remains.
Despite this, the paleontological evidence does not, generally corroborate molecular clock studies (contrary to the almost bizarre view expressed in Ayala et al., 1998). Bruce Runnegar 1982, p. 397) notes: "Few of the known late Precambrian animals have been closely related to Cambrian organisms, and none of the associated or coeval trace fossils has been thought to have been produced by the animals observed more directly. … What the trace fossil record does tell us, is that there were few large, mobile, bottom-dwelling animals before the end of the [Ediacaran]."
[Also see Xiao & Knoll 2000, top of p. 785a.]
"This explosion is perhaps the most striking single event documented by the fossil record. In the strict sense, the explosion refers to a geologically abrupt appearance of fossils representing all except two of the living [animal] phyla that had durable (easily fossilizable) skeletons. One of those two phyla is the Porifera sponges), which was present in the fossil record at an earlier time. The other is the Bryozoa, a phylum that contains some soft-bodied groups and may well have been present but not yet skeletonized. A number of enigmatic organisms of obscure relationships also appear during the explosion, enriching the early Cambrian fauna. Precision dating indicates that the explosion began at 530 Ma (million years ago) and ended before 520 Ma." Bowring et al. 1993).
|
The Tommotian and Atdabanian Ages (530 to 519 Ma)
The oldest known shelly fossils, simple tubes of the family Cloudinidae, first appear very near the end of the Proterozoic. Shelly fossils become more common and more diverse in the basal Cambrian – comprising the so-called 'small shelly fauna' – but this radiation is modest compared to the explosive diversification of Cambrian Epoch 2, a phenomenon which has come to be known as the Cambrian Explosion.
The "Small Shelly Fauna"
"Small shelly fossils near the base of the Cambrian mark the transition to a skeletonized fauna and the metazoan-dominated Phanerozoic fossil record. The recovery of articulated specimens composed of multiple sclerites discussed above, such as Wiwaxia, Halkieria and Microdictyon, suggests that much of the remaining "small shelly fauna" represent elements similarly employed as ectodermal armor in bilaterian Metazoa that have yet to be recovered in an articulated form. In addition, recent finds of cap-shaped shells composed of fused spicules of the early Cambrian age (Bengtson, 1992) support an argument of fusion of individual skeletal elements to form sclerites, plates, or shells (Haas, 1981).
"Thus, the engrailed data reported here, in combination with previous scenarios for the formation of ectodermal armor and recent fossil discoveries, suggest a singular evolution of invertebrate skeletons near the base of the Cambrian, followed by subsequent loss in several lophotrochozoan and ecdysozoan lineages. Such an interpretation, if substantiated, would have important implications for the Cambrian radiation, as it would constrain readily fossilizable exoskeletons to a single lineage, leading to a monophyletic clade of bilaterians. This would lead to a closer association of the bilaterian diversification with the base of the Cambrian." Jacobs et al. 2000: 345).
For a very long time, non-shelly fossils were unknown or poorly understood, the metazoan status of the Ediacarans was famously a matter of debate, and other lines of evidence had yet to be discovered. Thus, as far as anybody could be sure, the shelly fossil record was a literal record of metazoan evolution, and it seemed to be telling us that in Cambrian Epoch 2, animals diversified explosively from almost nothing to approximately the full range of basic archetypes known today, in as little as 10 million years.
However, "[t]he presence of true, though soft-bodied, triploblasts is now documented by worm burrows, by radular markings and body impressions of early mollusks, and by phosphatised embryos, of Ediacaran age." Seilacher et al. 1998: 80). Some, such as Kimberella and Parvancorina, can even be tentatively assigned to extant phyla. Nevertheless, although recent discoveries have greatly extended the record of sponges and bilateral animals, the earliest unequivocal paleontological evidence of metazoan life is no more than 600 Ma. Bromham et al. 1998).
Phosphatized Embryos
Reports of fossilized eggs and other early developmental stages of marine invertebrates are rare, probably mostly due to the difficulties of recognizing them. However, there is an abundance of small globular structures in the fossil record, leading up to and including that of the Cambrian.
-
Zhang and Pratt 1994 reported Middle Cambrian spherical fossils, 0.3 mm in diameter, that under a smooth membrane preserved a polygonal pattern which the authors interpreted as remains of blastomeres belonging to 64- and 128-cell stages of arthropod embryos. In some other cases, at least a general resemblance to eggs has been noted.
-
Bengtson & Zhao 1997) report two such occurrences of globular fossils from basal Cambrian rocks are eggs containing identifiable embryos of metazoans.
(A) 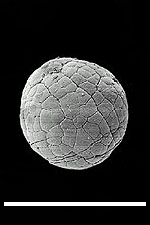 (B)
(B)
Fig. 1: (A) Reproduction of fig. 1A from Bengtson & Zhao 1997), a SEM image depicting a suggested metazoan embryo – possibly Olivooides multisulcatus – at approximately the 256-cell stage. Sample is NGMC (National Geological Museum of China) 9351 from the basal Cambrian Dengying Formation exposed in the Shizhonggou section, Shaanxi Province, China; scale is 500µm.
(B) Reproduction of fig. 1K from Bengtson & Zhao 1997), a SEM image of Olivooides multisulcatus. Sample is CAGS (Chinese Academy of Geological Sciences) 32372, also from the Dengying Formation exposed in the Shizhonggou section; scale is the same as A.
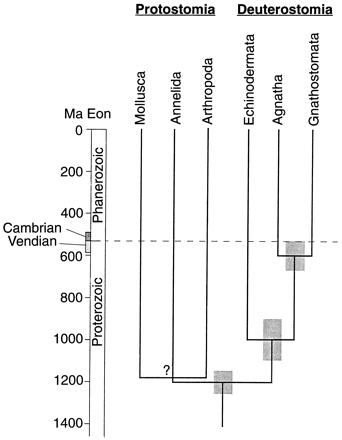
Whereas a literal interpretation of the Cambrian fossil record requires the near-simultaneous, 'late arrival' of nearly all metazoan phyla, recent genetic evidence reveals a different pattern, sometimes known as the 'slow burn' or 'early arrival' hypothesis. Age estimates derived from calibrated gene divergence studies tend to vary considerably today – the science is new – but a consistent pattern emerges, nevertheless. These studies all conclude that the major animal groups became separated from one another hundreds of millions of years before the Cambrian. Some studies (e.g. the classic Wray et al., 1996) place the age of the primary division of animals into protostomes and deuterostomes at around 1,200 Ma – much more than twice the age of the Cambrian Explosion.
Wray et al. (1996)
Genetic evidence has been used to suggest significant metazoan diversity far pre-dating the Ediacaran fossils (e.g. Wray et al., 1996: "Calibrated rates of molecular sequence divergence were used to test this hypothesis. Seven independent data sets suggest that invertebrates diverged from chordates about a billion years ago, about twice as long ago as the Cambrian. Protostomes apparently diverged from chordates well before echinoderms, which suggests a prolonged radiation of animal phyla.")
|
Fig. 1: Reproduction of fig. 2 from Wray et al. 1996 showing their estimated divergence times for selected metazoan phyla, based on seven genes, with standard errors indicated by shaded bars. The three estimated divergence times nest in agreement with well-corroborated phylogenetic relationships. Note that the chordate-echinoderm and chordate-protostome divergence times are significantly different from each other. Divergence times among the three protostome phyla were not estimated in their analysis. Although Wray et al. 1996 considers "the trend of the data to be more compelling than the exact numerical estimates" of divergence dates, the authors regard their conclusions as "incompatible with the Cambrian explosion hypothesis...." |
The Cambrian Explosion documents a real and dramatic radiation of skeletal morphologies, which might represent the parallel uptake of skeletonisation by numerous pre-existing, soft-bodied lineages or, alternatively, a rapid diversification within a relatively few already-skeletonized lineages. The resulting morphological disparity – as measured by the number of major animal types – was at least as great as the present. "Analysis of animal skeletons in relation to the multivariate, theoretical 'Skeleton Space' has shown that a large proportion of these options are used in each phylum. [The] structural elements deployed in the skeletons of Burgess Shale animals (Middle Cambrian) incorporate 146 of 182 character pairs defined in this morphospace. Within 15 million years of the appearance of crown groups of phyla with substantial hard parts, at least 80 percent of skeletal design elements recognized among living and extinct marine metazoans were exploited." Thomas et al. 2000: 1239). However, the species diversity produced by the Cambrian radiation, judged at the familial and generic levels, was much below that of succeeding periods there are more than four times as many marine families today).
 "Parvancorina minchami can be considered a good candidate in support of the notion that arthropods were present in the Ediacaran. Parvancorina appears to have had a tall, three-dimensional shape which has been flattened by subsequent compaction. The central axial ridge and the strongly arched anterior 'lobes' may be analogous to the midgut and gastric diverticulae which occur in the Burgess Shale arthropod Bugressia. About ten limbs appear to have been present, which may have been biramous. Scale bar in cms.
"Parvancorina minchami can be considered a good candidate in support of the notion that arthropods were present in the Ediacaran. Parvancorina appears to have had a tall, three-dimensional shape which has been flattened by subsequent compaction. The central axial ridge and the strongly arched anterior 'lobes' may be analogous to the midgut and gastric diverticulae which occur in the Burgess Shale arthropod Bugressia. About ten limbs appear to have been present, which may have been biramous. Scale bar in cms.
From Chris Nedin's Page
"The Early Cambrian metazoan radiation was accompanied by a marked diversification of organic-walled microorganisms. Early Cambrian protists include prasinophyte and dasyclad green algae, benthic foraminifers, and a diversity of acritarchs. Prokaryotes are represented by the carbonaceous and calcified remains of cyanobacteria, as well as by biogeochemical signatures, both isotopic and molecular." Knoll (1996: 51).
Duration
"A span of 40 million years embraces the appearance of the first small, simple shells that may have been secreted by metazoans and the subsequent exuberant diversity of Chengjiang and the Burgess Shale. This is not so short a time for an evolutionary 'explosion.' However, the proliferation of animals with well-differentiated hard parts characteristic of specific metazoan phyla was largely restricted to the last 15 million years of this interval." Thomas et al. 2000: 1239).
The application of absolute dates "to the boundaries of the Stages of the Lower Cambrian remains a difficult stratigraphic problem, but it is likely that the most critical Stages, the Tommotian and Atdabanian, are probably only 8-10 [million years] in duration; over 50 metazoan orders first appear in the [fossil] record during that interval (Valentine et al. 1991)." Valentine 1995: 89). As noted above, precision dating has now constrained the major Cambrian radiation to the interval 530 to 520 Ma (Bowring & Erwin 1998).
Genes or Skeletons?
Charles Darwin (1860) recognized that the sudden appearance of a diverse and highly derived fossil fauna in the Cambrian posed a problem for his theory of natural selection, "and may be truly urged as a valid argument against the views here entertained." Two obvious possibilities are that animal life did, indeed, evolve very abruptly about that time, or, alternatively, had existed long before, but that, for whatever reason, we have failed to find fossil evidence: the "late-" and "early-arrival" models, respectively.
The two patterns – Cambrian Explosion versus a very long (though obscure) metazoan history – are not necessarily incompatible, because they are based upon different criteria. The genetic evidence documents lines of descent and inheritance, irrespective of morphology, whereas the fossil record documents only the external expression of the genes.
Late-Arrival Models
The 'late arrival' model embodies a literal interpretation of the fossil record: that the evolution of the main animal groups took place both late in Earth's history, and as we now know, extremely rapidly.
What kind of mechanisms could have prompted such an accelerated pace of evolution? Various proposals have been advanced over the years, including:
-
A response to the evolution of Hox genes (Erwin et al. 1997).
-
A rise in macroscopic predation perhaps leading to an 'arms-race' style of evolution (Conway Morris 2000). Note, however, that the specific suggestion by Parker (2003) that such an arms race could have been fuelled by the acquisition of high-resolution vision in one or more groups, seems highly implausible (see the review by Conway Morris for a detailed critique).
-
Finally, perhaps we can look to the lifting of some external constraint such as insufficient atmospheric oxygen (Runnegar 1982; Brasier 1998, p. 548; Adouette et al. 2000, p. 4455).
Nowhere is the late-arrival model more eloquently discussed than in Stephen Gould's Wonderful Life (Gould, 1989), though nearly everyone would now agree that, in this book, Gould overstated his case by some orders of magnitude.
Early-Arrival Models
Darwin himself preferred the early-arrival explanation, noting that "before the lowest Silurian [the Cambrian system had not yet been recognized] stratum was deposited, long periods elapsed, as long as, or probably far longer than, the whole interval from the Silurian age to the present day; and that during these vast, yet quite unknown, periods of time, the world swarmed with living creatures." Darwin (1860).
Today we might regard Darwin's views as wonderfully prescient, for numerous Precambrian fossils have now indeed been collected, and modern techniques – impossible in Darwin's day – such as 'molecular clock' studies, strongly indicate metazoan evolutionary events having occurred deep within the Precambrian.
There can be little doubt, on the basis of trace evidence alone, that bilaterian metazoans existed in the Ediacaran, and possibly early in the Ediacaran. Although some traces are simple, rather featureless, winding trails, "others display transverse rugae and contain pellets that can be interpreted as of fecal origin. The bilaterian nature of these traces is not in dispute. Furthermore, such traces must have been made by worms, some of which had lengths measured in centimetres, with through guts, which were capable of displacing sediment during some form of peristaltic locomotion, implying a system of body wall muscles antagonized by a hydrostatic skeleton. Such worms are more complex than flatworms, which cannot create such trails and do not leave fecal strings." Valentine 1995: 90).
The Doushantuo Formation
The Doushantuo Formation, exposed near Weng'an in south central China, is of early Ediacaran age; ~590 Ma at its base to ~565 Ma at its top. Soft-tissue fossils preserving cellular structures provide strong evidence for a diverse biota predating by perhaps 5 million years (Ma) the earliest of the 'classical' Ediacaran faunas from Mistaken Point, Newfoundland, and existing a good 40 to 50 million years before the Cambrian explosion. Martin et al. 2000).
Earliest reports from the Doushantuo Formation include Xiao et al. 1998), which notes: "Embryos preserved in early cleavage stages indicate that the divergence of lineages leading to bilaterians may have occurred well before their macroscopic traces or body fossils appear in the geological record" (p. 553).
The documented biota now includes probable algae, sponges, cnidarians and bilaterians. Unfortunately, diagenetic effects are sometimes difficult to distinguish from genuine biological structures, so we can expect the significance of this evidence to be debated for some time to come yet.
(Read more about the Doushantuo Phosphate.)
Because several instances of this type of preservation have now been found, it may be that metazoan embryos are not uncommon as fossils but have simply been overlooked because of their minute size and nondescript morphology. If this is so it may provide us with the means to obtaining insight into the earliest stages of metazoan evolution; perhaps for establishing better paleontological dates for important divergence dates, and for reconciling genetic and fossil evidence.
"The lack of any evidence of horizontal burrowing in rocks older than about 575 Mya and of vertical burrowing in rocks older than 543 Mya is a strong argument that there existed no animals about 1 cm or longer that were capable of disturbing sedimentary layers before this time. When they do appear, these bilaterian traces indicate the presence of animals that had AP [anterior-posterior] differentiation, but there is no evidence of limbs" (Erwin & Davidson 2002, p. 3023, but note these authors were apparently unaware of Arenicolites).
 "Arenicolites is a simple U-shaped burrow oriented perpendicular to bedding. Different types of Arencolites can be interpreted on the basis of the breadth of the U. The generally accepted interpretation for Arenicolites is that it was a dwelling burrow."
"Arenicolites is a simple U-shaped burrow oriented perpendicular to bedding. Different types of Arencolites can be interpreted on the basis of the breadth of the U. The generally accepted interpretation for Arenicolites is that it was a dwelling burrow."
In preserving evidence of bilaterians, the Ediacaran record provides constraints on the protostome-deuterostome (P-D) divergence. If Kimberella is indeed a mollusc, as suggested by Fedonkin & Waggoner 1997, or certain trace fossils recorded from the Ediacara Hills and Zimnie Gory are correctly interpreted as radula scratches, we have evidence for derived protostomes at 555 Ma. Similarly, if Arkarua adami (from the Pound Subgroup, South Australia; Gehling 1987) is correctly interpreted as an echinoderm, we have evidence for a derived deuterostome of similar age. In either case, it follows that the protostome-deuterostome split must have occurred well before 555 Ma, which is consistent with most 'molecular clock' studies.
 Sets of paired hypichnial ridges strongly hint at an arthropod s.l. presence.
Sets of paired hypichnial ridges strongly hint at an arthropod s.l. presence.
The latter, however, mostly favour a P-D split far deeper in the Precambrian: some as early as 1,200 Ma (table 1). If correct, then the Cambrian explosion is clearly an artifact.