|
|
Bryophyta |
| Plants |
Bryophyta - 1 |
Bryophyta - 1
 If the mosses had not survived into the present, we would be forced to invent them as just the sort of intermediate we might expect between essentially aquatic algae and fully terrestrial plants. Mosses and, to a lesser extent, other basal plants, do have differentiated shoots. Although these are generally only a few millimeters tall, they are still designed to provide mechanical support against gravity on land -- the first such structure in any kingdom. Bryophytes also have leaves of a sort (phyllids). These are typically one cell thick and lack veins, although they may have a central thickening for support. Mosses also have rhizomes. These may have some function in extracting soil nutrients, although their primary function seems to be mechanical attachment to the substrate. Thus they are not true roots, but do approach that condition.
If the mosses had not survived into the present, we would be forced to invent them as just the sort of intermediate we might expect between essentially aquatic algae and fully terrestrial plants. Mosses and, to a lesser extent, other basal plants, do have differentiated shoots. Although these are generally only a few millimeters tall, they are still designed to provide mechanical support against gravity on land -- the first such structure in any kingdom. Bryophytes also have leaves of a sort (phyllids). These are typically one cell thick and lack veins, although they may have a central thickening for support. Mosses also have rhizomes. These may have some function in extracting soil nutrients, although their primary function seems to be mechanical attachment to the substrate. Thus they are not true roots, but do approach that condition.
The bottom line is that, structurally, mosses really differ from rhyniophytes in only one aspect: mosses lack the particular, specialized vascular tissues of tracheophytes. That alone is sufficient to explain the lack of big leaves, long stems, and true roots. This whole complex of characters is thus probably primitive. The other distinctive character of mosses is that the plant we normally observe is the haploid, gametophyte stage. But this character is shared with liverworts (basal embryophytes) and so is also probably plesiomorphic.
Curiously, in hornworts (also basal embryophytes) the sporophyte generation is dominant. In addition, it turns out that the leaves of moss probably evolved independently from the leaves of higher plants. So the relationships of the mosses and basal embryophytes are still uncertain. What really seems to set mosses apart is their unique form of leaf. What may unite mosses with higher plants is (a) the presence of stomata to control water loss and (b) meristem apical growth) in the sporophyte generation. See, Friedman et al. (2004). However, even these similarities may not be strictly homologous. Phylogenetically, we treat Bryophyta as Moss > Quercus.
A word about terminology. "Bryophyta" was formerly used, and is still used by many to include the liverworts and hornworts, as well as the mosses. Since the branch order and monophyly of the mosses and the two liverworts (Marchantiophyta) and hornworts (Anthocerotophyta) remains unclear, this is understandable. However, we prefer the more restrictive use of the term to mean just mosses, since the mosses are probably monophyletic. The wort + moss group, however structured, is almost certainly paraphyletic. That is, the broader definition takes in all of the basal radiation of embryophytes (land plants), including the direct ancestors of the "higher plants."
Image: moss on rocks in Iceland from
Arrakeens Site.
 As mentioned, the gametophyte (haploid) generation is dominant in the life cycle of mosses. The sporophyte (diploid) form remains attached to and dependent on the gametophyte at all times. That being the case, we can best describe the mosses starting from the bottom and working up to the gametic structures, then proceeding from the archegonium (ovary) to the developing sporophyte as if it were an extension of the gametophyte, which is essentially the case. There is considerable variety among mosses; but, when in doubt, we sometimes defer to the peat moss Sphagnum, which is not only the most basal living moss, but also the most common. (Unfortunately, it is also, in some respects, quite atypical. Shaw et al. (2003))
As mentioned, the gametophyte (haploid) generation is dominant in the life cycle of mosses. The sporophyte (diploid) form remains attached to and dependent on the gametophyte at all times. That being the case, we can best describe the mosses starting from the bottom and working up to the gametic structures, then proceeding from the archegonium (ovary) to the developing sporophyte as if it were an extension of the gametophyte, which is essentially the case. There is considerable variety among mosses; but, when in doubt, we sometimes defer to the peat moss Sphagnum, which is not only the most basal living moss, but also the most common. (Unfortunately, it is also, in some respects, quite atypical. Shaw et al. (2003))
Mosses manage to look like small tracheophytes, but behave more like liverworts. So, for example, mosses have small, threadlike basal processes which look like roots. In fact, these are simply rhizoid holdfasts, not very different from those found in algae. It is likely that the rhizoids absorb water, but only because almost everything in a moss absorbs water. However, a moss lacks a highly specialized vascular system, so the water is usually stored in the ubiquitous moss water storage cells. From these cells, water probably diffuses, very slowly, to other parts of the plant; but, in most cases, the probable function of the water storage cell is structural. Since mosses have no wood, either, they use the turgor pressure of these storage cells to satisfy much of their modest need for structural support. Another common moss strategy for structural support is to grow very densely.So, like academic communities, they grow progressively larger and denser, maintaining their continued existence by sheer bulk without having any real roots in the environment which supports them.
 Actually, we may further extend this dubious simile, since mosses have likewise developed elaborate designs for clinging to each other, so that the entire mass behaves like a foam rubber pillow -- conforming to the slightest pressure, but springing back to business as usual when the pressure is removed. One such strategy is the use of yet more rhizoids, growing from the shoot, which entangle adjacent shoots and their rhizoids.
Actually, we may further extend this dubious simile, since mosses have likewise developed elaborate designs for clinging to each other, so that the entire mass behaves like a foam rubber pillow -- conforming to the slightest pressure, but springing back to business as usual when the pressure is removed. One such strategy is the use of yet more rhizoids, growing from the shoot, which entangle adjacent shoots and their rhizoids.
Sakakibara et al. (2003) have shown that rhizoids on gametophores of Physcomitrella differentiate in response to the ubiquitous plant hormone auxin, followed by a (presumably more specific) signal from HD-ZIP homeodomain genes. This is essentially the same mechanism which is involved in the development of roots in higher plants. Id.. However, before you get excited, note that it is also the same mechanism involved in, e.g. the specification of leaf morphology in higher plants. Micol & Hake (2003). It appears to be a general mechanism for developing lateral structures. Still, this all encourages us to think that there is a broad, underlying homology between roots and rhizoids.
Mosses generally have a well-defined shoot in the gametophyte generation. In early development, the plant may take a flattened form, suspiciously similar to the thallus of an alga or liverwort. More typically, the spore develops a long thread-like, recumbent growth, the protoneme, which in turn buds off lateral caulonemata which develop into stem-like gametophore structures on which the gametes will develop in a terminal gametangium. This structure typically has a well-developed pattern of leaves, as in the image of Physcomitrella above. It is common to see this referred to as a "stem" or "shoot" although it has no xylem or phloem and is therefore not really similar to an axis of a vascular plant.
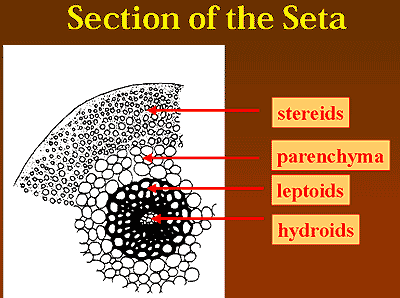 Rather than the xylem or phloem of tracheophytes, mosses have tissues made up of hydroid and leptoid cells. These are frequently referred to as "vascular tissues," and they do probably serve analogous functions. The subject has been reviewed by Ligrone et al. (2000). However, they are not lignified. The hydroid tissues, in particular, are the general run of moss water storage cells and are probably only remotely related to the xylem of tracheophytes. Leptoids are not as well understood, but might be homologous to phloem. However there is great variation in the structure and degree of specialization of these tissues, and their phylogenetic distribution is somewhat unexpected. Unfortunately, we have to break off with these vague and enigmatic generalities, or we will never get on with the rest of the job. As it turns out, most water and nutrient transport is actually accomplished external to the shoot, using a variety of rather ad hoc tools -- small channels on the surface, gutters and catchments formed by leaves, playing games with mucous and surface tension, etc.
Rather than the xylem or phloem of tracheophytes, mosses have tissues made up of hydroid and leptoid cells. These are frequently referred to as "vascular tissues," and they do probably serve analogous functions. The subject has been reviewed by Ligrone et al. (2000). However, they are not lignified. The hydroid tissues, in particular, are the general run of moss water storage cells and are probably only remotely related to the xylem of tracheophytes. Leptoids are not as well understood, but might be homologous to phloem. However there is great variation in the structure and degree of specialization of these tissues, and their phylogenetic distribution is somewhat unexpected. Unfortunately, we have to break off with these vague and enigmatic generalities, or we will never get on with the rest of the job. As it turns out, most water and nutrient transport is actually accomplished external to the shoot, using a variety of rather ad hoc tools -- small channels on the surface, gutters and catchments formed by leaves, playing games with mucous and surface tension, etc.
One more cell type worth mention is the stereid. These are thick-walled cellulosic support cells found near the perimeter of the shoot (and also in leaves and setae) which presumably function as rebar. They are easily seen on the perimeter of the shoot in the cross-section of Neckera in the image above. Growth and differentiation of the shoot, and of nearly everything else in mosses, is driven by a single apical cell.
Image: Seta cross-section from the course web site of Prof. Robert S. Eagan, University of Nebraska at Omaha (who has an excellent pdf on bryophyte anatomy).
Leaves & Peripheral Structures
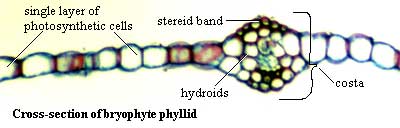 Bryophyte leaves are generally arranged in a very stereotyped way around the shoot, typically a spiral with four positions (e.g. Physcomitrella). The bryophyte leaf typically consists of a single layer of photosynthetic cells with a midline vein, rib, nerve or costa (all the same thing) containing hydroids and/or rows of stereids for mechanical support. Botanists who prefer a more exacting definition of leaf refer to them as phyllids, since moss leaves lack many of the advanced features of tracheophyte leaves. The polytrichid mosses may have much more elaborate structures in both leaves and shoot. By contrast, many mosses lack even the customary costal thickening.
Bryophyte leaves are generally arranged in a very stereotyped way around the shoot, typically a spiral with four positions (e.g. Physcomitrella). The bryophyte leaf typically consists of a single layer of photosynthetic cells with a midline vein, rib, nerve or costa (all the same thing) containing hydroids and/or rows of stereids for mechanical support. Botanists who prefer a more exacting definition of leaf refer to them as phyllids, since moss leaves lack many of the advanced features of tracheophyte leaves. The polytrichid mosses may have much more elaborate structures in both leaves and shoot. By contrast, many mosses lack even the customary costal thickening.
 In addition to leaves, moss stems may bear structures for aesexual reproduction -- gemmae or bulbils. These are not complex. Mosses have great powers of regeneration and can regenerate an entire plant from an isolated segment of a shoot. The gemmae therefore consist of a small ball of undifferentiated cells. Finally, mosses may, like higher plants, also have a waxy cuticle and stomata to help control evaporation. The stomata are restricted to the sporophyte generation -- not only in mosses, but in all land plants, with the exception of the extinct polysporangiophytes -- and many mosses lack stomata entirely. Nevertheless, general statements to the effect that mosses lack cuticles and/or stomata are simply incorrect. See, generally, review by Raven 2002).
In addition to leaves, moss stems may bear structures for aesexual reproduction -- gemmae or bulbils. These are not complex. Mosses have great powers of regeneration and can regenerate an entire plant from an isolated segment of a shoot. The gemmae therefore consist of a small ball of undifferentiated cells. Finally, mosses may, like higher plants, also have a waxy cuticle and stomata to help control evaporation. The stomata are restricted to the sporophyte generation -- not only in mosses, but in all land plants, with the exception of the extinct polysporangiophytes -- and many mosses lack stomata entirely. Nevertheless, general statements to the effect that mosses lack cuticles and/or stomata are simply incorrect. See, generally, review by Raven 2002).
Image: phyllid cross-section from website of Dr. George P. Chamuris, Bloomsburg University.
Mosses may be either monoecious hermaphroditic) or dioecious. Similar species within the same genus may differ in this character, and there is little phylogenetic consistency. The gametangia are normally located in terminal clusters at the end of the shoot. However, in Takakia and Sphagnum, generally considered two of the most basal mosses, gametangia are associated with leaves along the shoot. As we might expect in such a regime, the male and female gametangia have somewhat similar structures. See the image from Alejandro 2004). Notwithstanding their general similarity, the archegonia of mosses differ externally from antheridia in having a longer neck and longer stalk. The neck encircles a tube which leads to the cavity (venter) containing the female gamete.
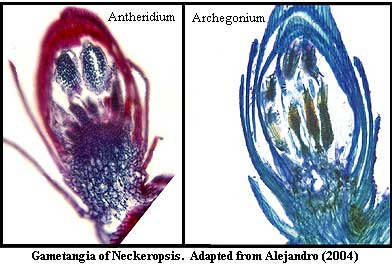 Both gametangia form from a stereotyped series of divisions of a terminal apical cell, unlike the gametangia of liverworts and hornworts. However, once the initial division are complete, the developmental pattern of the archegonium is the same in all three groups. "In all three groups, the process involves three longitudinal divisions that form a central triangular axial cell surrounded by three peripheral cells. The peripheral cells form the neck and venter while the axial cell gives rise to the neck canal cells, ventral canal cell and egg. Further divisions in the peripheral cells typically result in a neck of five or six cell rows." Renzaglia et al. (2000: 775).
Both gametangia form from a stereotyped series of divisions of a terminal apical cell, unlike the gametangia of liverworts and hornworts. However, once the initial division are complete, the developmental pattern of the archegonium is the same in all three groups. "In all three groups, the process involves three longitudinal divisions that form a central triangular axial cell surrounded by three peripheral cells. The peripheral cells form the neck and venter while the axial cell gives rise to the neck canal cells, ventral canal cell and egg. Further divisions in the peripheral cells typically result in a neck of five or six cell rows." Renzaglia et al. (2000: 775).
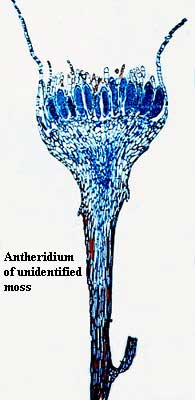
The following description of the structure and function of the antheridium from Phylum Bryophyta is better than anything we could come up with, so we quote it in extenso: "Antheridia are equally elongated with a long narrow stalk and banana-shaped antheridial body. The sterile jacket is one layered except at the tip where an operculum forms. When mature, the operculum cells dissociate and release blocks of spermatozoids individually enclosed in "vesicles". Spermatozoids are biflagellated [sic], coiled and thread-like [and, we might add, absolutely bizarre in both development and appearance]. Transport of spermatozoids may be effected by rain droplets encountering the cup-like antheridial shoot and splashing packets of cells to adjacent plants." Interestingly, this method of dispersal, by raindrops agitating a cup-like structure, is essentially the same as the method used to disperse gemmae during aesexual reproduction in both moss and other basal plants. Compare the very different dispersal method for spores, discussed on the next page.
Credits: the Phylum Bryophyta page quoted above is part of Land Plants On Line, the product of Dan Nickrent and Karen Renzaglia both well-known botanists) of the University of Southern Illinois at Carbondale. Antheridium image from the Biology 211 Lab site of Iowa State University.
uploaded ATW050928
modified ATW090227
 If the mosses had not survived into the present, we would be forced to invent them as just the sort of intermediate we might expect between essentially aquatic algae and fully terrestrial plants. Mosses and, to a lesser extent, other basal plants, do have differentiated shoots. Although these are generally only a few millimeters tall, they are still designed to provide mechanical support against gravity on land -- the first such structure in any kingdom. Bryophytes also have leaves of a sort (phyllids). These are typically one cell thick and lack veins, although they may have a central thickening for support. Mosses also have rhizomes. These may have some function in extracting soil nutrients, although their primary function seems to be mechanical attachment to the substrate. Thus they are not true roots, but do approach that condition.
If the mosses had not survived into the present, we would be forced to invent them as just the sort of intermediate we might expect between essentially aquatic algae and fully terrestrial plants. Mosses and, to a lesser extent, other basal plants, do have differentiated shoots. Although these are generally only a few millimeters tall, they are still designed to provide mechanical support against gravity on land -- the first such structure in any kingdom. Bryophytes also have leaves of a sort (phyllids). These are typically one cell thick and lack veins, although they may have a central thickening for support. Mosses also have rhizomes. These may have some function in extracting soil nutrients, although their primary function seems to be mechanical attachment to the substrate. Thus they are not true roots, but do approach that condition.
 Actually, we may further extend this dubious simile, since mosses have likewise developed elaborate designs for clinging to each other, so that the entire mass behaves like a foam rubber pillow -- conforming to the slightest pressure, but springing back to business as usual when the pressure is removed. One such strategy is the use of yet more rhizoids, growing from the shoot, which entangle adjacent shoots and their rhizoids.
Actually, we may further extend this dubious simile, since mosses have likewise developed elaborate designs for clinging to each other, so that the entire mass behaves like a foam rubber pillow -- conforming to the slightest pressure, but springing back to business as usual when the pressure is removed. One such strategy is the use of yet more rhizoids, growing from the shoot, which entangle adjacent shoots and their rhizoids.


 Both gametangia form from a stereotyped series of divisions of a terminal apical cell, unlike the gametangia of liverworts and hornworts. However, once the initial division are complete, the developmental pattern of the archegonium is the same in all three groups. "In all three groups, the process involves three longitudinal divisions that form a central triangular axial cell surrounded by three peripheral cells. The peripheral cells form the neck and venter while the axial cell gives rise to the neck canal cells, ventral canal cell and egg. Further divisions in the peripheral cells typically result in a neck of five or six cell rows."
Both gametangia form from a stereotyped series of divisions of a terminal apical cell, unlike the gametangia of liverworts and hornworts. However, once the initial division are complete, the developmental pattern of the archegonium is the same in all three groups. "In all three groups, the process involves three longitudinal divisions that form a central triangular axial cell surrounded by three peripheral cells. The peripheral cells form the neck and venter while the axial cell gives rise to the neck canal cells, ventral canal cell and egg. Further divisions in the peripheral cells typically result in a neck of five or six cell rows."