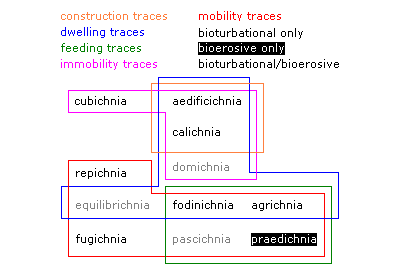
Fig. 1: Diagram illustrating the nature of the eleven established ethological trace fossil classes, and ichnoguilds within which they can be included (modified from Gibert et al. 2004).
| Palaeontology | ||
| Life | Palaeontology |
| Lagerstätten | ||
| Evolution | Life | Systematics |
Life |
Paleontology |
Trace fossils are indirect evidence of life in the rock record. There are several fundamental differences between traces and body fossils, which lead to their unique classification and nomenclature. In general they make poor zone fossils, but they are powerful tools in palaeoenvironmental interpretation. Studies show various general trends in their diversity and range, throughout the Phanerozoic. There are several sites around the world where a wide range and diversity can be observed.
Trace fossils, or ichnofossils (from the Greek ikhnos meaning "track" or "trace"), are those structures and details preserved in rocks that provide indirect evidence of life in the past, or indeed "traces" of it. The most familiar of these are the tracks, trails, burrows, gastroliths, coprolites, impressions, borings, etc., made by invertebrates of all phyla (Pickerill 1994), but craniates, plants, fungi, and bacteria also contribute significantly to their number.
Ichnofossils are found on and within both hard and soft substrates, especially in sandstones or between two contrasting lithologies (Allaby & Allaby 1999). They constitute an alternative fossil record (Crimes & Droser 1992) from the more familiar physical occurrences of the organisms themselves, i.e. body fossils (bones, shells, carbonised leaves, teeth, etc.) and, although they may be visually less spectacular and inspiring at first glance, they are just as important. On the surface of it there is a considerable overlap between these two fossil realms, both in the information they can yield and in the way they might be studied. However, as one delves in more detail into this area of palaeontology, it becomes clear that traces differ in nature substantially from the traditional fossil type, and should be studied as such, with their own specific schools of thought.
The study of trace fossils, ichnology, can be divided into two distinct fields: palaeoichnology (ancient, fossilised traces) and neoichnology (modern traces created by extant organism groups). This is a subdivision of convenience - it is not to say that a palaeoichnologist would have nothing to do with modern traces or vice versa, it is quite the opposite - the two areas are linked inextricably. In fact, this link is one of the few occasions where the principle of uniformitarianism is turned on its head. Lebensspuren such as traces are much easier to study and much better known in fossil form than they are when freshly created in soft sediments today, therefore in this case "the past is the key to the present". This converse property is one of the major differences identified by Adolf Seilacher (1967) between trace and body fossils. The other differences, as well as many of the major trace fossil properties and points of contention in ichnology today, are discussed below.
The morphology of trace fossils is controlled predominantly by the behavioural (ethological) characteristics of the organisms producing them (eg Osgood, 1970; Häntzschel 1975). The physical dimensions of the organisms also influence most traces to a degree, but usually to a far lesser extent. With this fact in mind, it becomes clear why ichnologists have a hard time identifying the organism that produced a particular trace. Indeed, traces are perhaps the most vulnerable biogenic structures to confusion with pseudofossils (inorganic structures, impressions, or markings that bear resemblances to true fossils) by even the most experienced of palaeontologists, especially in such cases where they are known to occur in the same sequence (Benton & Gray 1981). There have been several such misdiagnoses in the past, prompting Bromley (1990) to note how: "We have inherited a set of names for trace fossils the most venerable of which originated in a series of misconceptions and misidentifications." Pickerill (1994) provides several examples of dewatering structures assuming such ersatz roles, with various forms morphologically mirroring ichnospecies such as Dactyloidites ottoi Geinitz 1849, Arenicolites variabilis Fürsich 1974c, and Skolithos linearis Haldeman 1840, among others. Therefore great care and attention to detail is often required before one can even verifiably assign trace fossil status to a structure, let alone identify its progenitor.
Once a trace is confirmed, there are several complicating factors that must be considered before and serious effort can be made to verify which organism produced it. Frey & Seilacher (1980) highlighted many of these, including: (1) different behaviours within an individual organism may result in the creation of different traces, so one trace maker may be responsible for two or more trace fossil species; (2) on the other hand, similar behaviours between taxonomically unrelated organisms (possibly members of different phyla) may produce very similar traces, an excellent example being Chondrites von Sternberg 1833, which was originally described as plant remains but is now thought to have been created by deposit-feeding animals as they combed the sediment in search of food; (3) the ontogeny of the trace may not necessarily be related to the ontogeny of its maker, ie, as the organism develops, the traces it creates may or may not develop in a similar way (size, relative dimensions, shapes, etc); and (4) a single trace fossil type might represent a number of different behavioural traits.
An additional complexity that is often overlooked is the fact that animals are not machines. Slight variations from one day to the next in the way an organism burrows, for example, might throw up slightly different traces, all reflecting exactly the same behaviour in the same organism. Causes of this might be changes to environmental conditions on a local scale, varying sediment composition and consistency, or there may be no particular factor, just random variations. Furthermore, a trace created by the same organism as a result of the same activity may look quite different if preserved under different depositional and/or diagenetic conditions (Seilacher 1964). These factors may lead to the recognition of separate ichnospecies under the same ichnogenus, when really they should be considered as the same trace. Traces may also occur as the result of a combination of the behaviours of two separate organisms, producing a single structure (Brenchley & Harper 1998).
For these reasons, it is rare for a trace fossil to be assigned to a particular organism or even a higher taxon, with the exception of certain vertebrate and arthropod repichnia (Seilacher 1964, 1967a). Usually, however, it can be determined whether the trace is of metazoan, plant, bacterial, or fungal origin.
An example of a trace that has been assigned to a specific organism is that of a recently discovered eurypterid trackway created by a species of Hibbertopterus. This trace is a good example of how ichnofossils can provide valuable information that cannot be gleaned from body fossils. If the interpretation is correct, it shows that giant water scorpions were able to survive out of water for at least short periods 330 Ma ago in the Carboniferous, which could only be speculated until now. It is one of the largest traces of its type known, thought to have been created by the giant invertebrate moving along a soft, sandy beach (Whyte 2005).
When considering the immense difficulties in identifying the creator of a biogenic structure, it is hardly feasible to attempt to systematically group their traces into any kind of a phylogeny. Two classification systems have prevailed over the past few decades: (1) the ethological classification scheme proposed by Seilacher (1953, 1964); and (2) the toponymic categorization offered by Martinsson (1970).
The standard suffix used in the technical terminology of trace fossil types is -ichnion (plural -ichnia).
Even if there can be no authoritative taxonomic definition for a trace, there is usually strong evidence for the type of behaviour that produced it (Seilacher 1964). This was the main factor in motivating Seilacher to propose a classification scheme based on behaviour. There are several criteria that can give a trace away as being ethological in origin, many of which are addressed in detail by Ekdale et al. 1984 and Donovan 1994. For example:
Seilacher based his system upon five major groups: (1) cubichnia, formed on the top of a soft sediment by a resting or hiding animal; (2) domichnia, which reflect the life position of the organism; (3) fodinichnia, burrows found in soft sediments caused by feeding behaviour; (4) pascichnia, grazing traces leaving evidence of an animal scouring a surface for food; and (5) repichnia, which are the impressions of an animal track.
Cubichnia are generally the most morphologically influenced of the ethological trace groups, reflecting the size and shape of the trace maker. Arthropods, cnidarians, and echinoderms are responsible for the majority of them (Brenchley & Harper 1998). Perhaps the most famous examples are Asteriacites von Schlotheim 1820, thought to be made by an asterozoan, and Rusophycus Hall 1852, created by a trilobite or similar arthropod.
Domichnia are dwelling structures, principally created by infaunal filter feeders, usually in the form cylindrical burrows. They reflect the life position of the organism, which would have remained there until death or some external stimulus caused it to move on. They are most often vertical (e.g. Skolithos Haldemann 1840) but some are horizontal or inclined to the bedding strata (Brenchley & Harper 1998), akin to the burrows made by spatangoids. There are many well-known examples, including Arenicolites Salter 1857, and Thalassinoides Ehrenberg 1944.
Fodinichnia, such as Teichichnus Seilacher 1955, represent the three-dimensional feeding patterns of deposit feeders as they scour the sediment, picking up any nutriment contained within (Bromley 1994). Typically the trails created in this way are phobotactic (i.e. they do not overlap or repeat), since the most efficient way for a deposit feeder to acquire maximum nutrition is by not crossing the same area more than once. Chondrites von Sternberg 1833 and Rhizocorallium Zenker 1836 are other examples.
Pascichnia, like fodinichnia, represent feeding behaviour, but are made by a different trophic group – grazers. They are associated principally with hard, mineral substrates, and are concentrated on surfaces rather than in a 3-dimensional environment, therefore bearing little resemblance to endogenic, soft-sediment fodinichnia. A wide variety of patterns result from this grazing activity, from spirals to sinuous furrows, and phobotaxis is again a feature, previously formed branches rarely crossing (Brenchley & Harper, 1998). Cosmorhaphe Fuchs 1895 is an example.
Repichnia are locomotory tracks interpreted as evidence of an organism's direct movement from one point to another (Seilacher 1964) on or within soft sediment. This interpretation is usually based on the fact that the track is more or less straight, or with a slight curvature. This group constitutes perhaps the most wide-ranging and best-known traces, from dinosaur footprints (e.g. Anomoepus E. Hitchcock 1848) to arthropod trackways (e.g. Cruziana d'Orbigny 1842), and even including early human footprints, such as those at Laetoli (fig. 3).
Since Seilacher's pioneering paper, several other ethological trace groups have been recognised, and including Seilacher's original five, eleven are currently accepted by the ichnological community (Bromley 1996), with several others of tentative status. The other currently accepted groups are as follows:
The other, more tentative, groups (e.g. the polychresichnia, suggested by Hasiotis 2003) are shown in table 1. They tend to be subsets of this core set of eleven ethological classes. An exception to this might be fixichnia which, as defined by Gibert et al. 2004, cannot be fully accommodated in any one of the more established classes.
| Group | Subgroup | Definition | Status | |
|---|---|---|---|---|
Metazoan Traces |
aedificichnia |
traces formed from structures built above the substrate and cemented in place by an organism. |
accepted | |
| calichnia |
structures that were used for breeding purposes, e.g. bee cells or any of various nests. |
accepted | ||
| agrichnia |
networks of burrows designed to catch migrating meiofauna or culture bacteria within. |
accepted | ||
| chemichnia |
agrichnia that are specifically designed to culture bacteria for harvesting. |
tentative | ||
| fodinichnia |
burrows created by deposit feeders (e.g. nematodes) as they scoured the sediment for nutriment. |
accepted |
||
| pascichnia |
patterns on the surface of a substrate (esp. a hard substrate) that reflect the feeding patterns of grazing organisms. |
accepted |
||
| praedichnia |
evidence of predatory behaviour, e.g. bite marks on bones, shell borings, etc. |
accepted |
||
| mordichnia |
praedichnia that show evidence of the death of an organism after it has been predated. |
tentative | ||
| cubichnia |
impressions left on the surface of a soft substrate that show where an organism was resting or hiding. |
accepted | ||
| domichnia |
traces, e.g. burrows, that reflect the life position of the organisms that created them. |
accepted | ||
| fixichnia |
the superficial etchings left after a sessile organism has used a soft or skeletal body part to anchor itself to a mineral substrate. |
tentative | ||
| equilibrichnia |
traces showing the response of an organism to accommodate varying sedimentation rates, e.g. U-shaped burrows with spreiten. |
accepted | ||
| fugichnia |
sediment disruption caused by an organism attempting to escape a sudden burial from increased sedimentation. |
accepted | ||
| taphichnia |
fugichnia showing an unsuccessful attempt to escape burial, which resulted in the organism's death. |
tentative | ||
| repichnia |
ichnofossils created by an organism moving from one station to another in a definite direction. |
accepted | ||
| cursichnia |
track traces that reflect locomotory behaviour in the form of crawling, walking, etc in the organism that created them. |
tentative | ||
| natichnia |
repichnia that were caused by natatorial behaviour, e.g. a fish disturbing soft sediment by swimming low over it. |
tentative | ||
| volichnia |
impressions (chiefly of insects) that show the position in which a flying organism landed on a soft sediment. |
tentative | ||
polychresichnia |
trace fossils caused by multiple behaviours, e.g. calichnia also serving as domichnia, or cubichnia caused by ambush predators. |
tentative | ||
Metaphytan traces |
cecidoichnia |
abnormal outgrowths of plant tissues that are produced in response to invasion by insect parasites, viruses, bacteria, and fungi. |
tentative |
|
| corrosichnia |
rhizocretions arising because of the corrosive action of plant roots. |
tentative |
||
| sphenoichnia |
bioturbation that reflects root growth growth and spreading down into a soft sediment. |
tentative |
||
| Table 1: Ethological ichnofossil groupings and characteristics. | ||||
A further proposal, put forward by Mikuláš 1999, is the idea of distinguishing traces created by metazoans from those of metaphytan origin. He identified three separate classes relating to plant traces: corrosichnia and sphenoichnia being those rhizoliths caused by the action of roots corroding and bioturbating lithic sediments respectively; and cecidoichnia, in which animal-plant (rarely plant-plant, fungus-plant or moneran-plant) interactions leave a gall on a xylic substrate (i.e. the plant itself). Little work has been subsequently added to this area of ichnology but it remains an intriguing prospect.
It is possible to further classify ethological groups into the bioerosive and bioturbational realms, according to the nature of the substrate. Bioerosion represents the actions of animals, plants, and microbes that sculpt and penetrate hard substrates (Bromley 1994). Bioturbation occurs in soft sediments, where a range of behaviours can disrupt the grains and leave patterns that are subsequently preserved upon lithification. When one analyzes each trace-producing behavioural trait in this regard, it becomes clear that a wider ethological diversity of traces is produced within soft sediments than hard substrates (Gibert et al. 2004). As fig. 1 shows, most of the accepted ethological groups are exclusively bioturbational, praedichnia being the only exclusively bioerosive class. Furthermore, of those groups overlapping between the two, only pascichnia is found more commonly on hard substrates.
The groupings in Fig. 1 are somewhat superficial and do not constitute viable ichnotaxa or, for the most part, ichnotaxobases. They are loosely-defined "ichnoguilds" based on their sharing of particular resources or behaviour, and provide nothing more than a general idea of which classes are more closely related to others. For example, it can be seen at a glance that cubichnia only share one characteristic with three other groups, in being made by an organism that was not moving around at the time. On the other hand, fodinichnia and agrichnia share facets of organismal behaviour with all classes except cubichnia, in being made by mobile organisms that were feeding, and may also have used this feeding area as a life site.
|
Fig. 1: Diagram illustrating the nature of the eleven established ethological trace fossil classes, and ichnoguilds within which they can be included (modified from Gibert et al. 2004). |
The toponomic (or morphotopographic) system provides a completely different way to consider trace fossils, being more or less unrelated to the organisms producing them. The idea is to look at where the traces occur in relation to the casting medium, since an important consideration when classifying traces is how its morphological expression can be influenced by preservation processes (Bromley 1990). The most common environment for trace fossil observation is on the tops and soles of sandstones, a feature that indeed triggered the introduction of several stratinomic terms in this regard, which has prompted several authors to organise them into classification systems (Bromley 1990).
The first attempt at classifying traces in this way was by Seilacher (1953), in a system he subjected to subsequent revision and modification (1964a, 1964b). He split traces into two basic types, those in full relief ("Vollformen", in which the full structure is preserved wholly within a sediment) and those in semirelief (or "Halbformen", traces preserved at an interface between two strata). Full reliefs can only be created by infaunal animals, reflecting their behaviour within the sediment, and have two main forms: fills, which contain sediment of the same nature as the surrounding medium or perhaps some other lithology; and cavities, which are open burrows. Several ways in which semireliefs can form were also detailed, with this category further split into boundary reliefs and cleavage reliefs. Boundary reliefs are those traces that occur strictly at the contact between two beds, reflecting reworking of the sediment by the animal at the upper or lower interface immediately before preservation. Boundary reliefs found on the upper surface of the substrate are called epireliefs, with those on the soles of strata termed hyporeliefs. Cleavage reliefs show deformation of subsurface laminae, before some subsequent event such as erosion has brought the trace to a stratal boundary, and as such an individual specimen may resemble a true boundary relief. Most of these trace categories are heterogeneous in nature.
In addition to these descriptive classes Seilacher also identified a genetic basis for categorisation, introducing three terms to this end: endogene, meaning the trace is filled with material of the same origin as the host sediment; exogene, used to describe traces with a composition alien from the casting medium due to coverage by a different sediment; and pseudexogene, describing the scenario whereby a structure is actively filled by the organism with a material other than the surrounding sediment (e.g. a burrow with a mud lining).
Simpson (1957) introduced an alternative scheme, perhaps a little less aesthetic, with poorer organisation and more ambiguity due to its openness to interpretation. He recognised four distinct categories for all trace fossils to fall within regarding the nature of their preservation:
The most recent of the major toponomic classifications has been provided by Chamberlain (1971). His system more or less regurgitated previous work, but it is a very simple scheme in which traces can be placed in one of three categories: (1) endogene traces, which are structures filled within the host sediment, and therefore are homogeneous with the casting medium. The sediment infilling may have occurred either actively (by the organism itself) or passively (by some other process, after the organism has created the structure); (2) epigene traces, which are traces produced on the upper surface of a sediment then preserved by being quickly covered by a sediment of a different lithology; (3) intergene traces, which are endogenic structures formed at a lithological interface or the junction between two beds.
Perhaps the most authoritative and widely-used toponomic system, however, is that proposed by Martinsson (1965, 1970). He identified four ichnofossil groups with the morphotopographic basis: (1) endichnia, which are found within the casting medium; (2) epichnia, ridges or grooves found on the upper surface of the bed; (3) exichnia, traces found within a medium other than the sediment of origin; and (4) hypichnia, which are basically the opposite of epichnia, being ridges or grooves on the underside of the bed. This is again very similar to the Seilacherian system, as well as Chamberlain's offering, but is broadly regarded as the most aesthetic and all-encompassing of the schemes, although Seilacher's system would certainly seem the most thorough. The two are compared in Fig. 2, a classic diagram found in many works (e.g. Bromley 1990; Brenchley & Harper 1998) showing the parallels between the two most popular schemes.
The ambiguities faced by ichnologists in determining the creator of a trace means that the naming of trace fossils needs to have its foundations on completely different principles from that of more familiar, organismal taxonomy which is more or less based on phylogeny. In 1961, the International Committee for Zoological Nomenclature, having regarded the various complexities assigned to trace fossil nomenclature and its inconsistencies with established codes for animals and plants, ruled that any name proposed after 1930 would be no longer available. Traces described after this time therefore had no legal status, but those that were already established did. This action led many to propose a new, separate nomenclatural code specifically for trace fossils (e.g. Sarjeant & Kennedy 1973). There were problems with this, however (see Pemberton & Frey 1982 for fuller discussion), and in 1985 trace fossil names were again brought under the provisions of the International Code of Zoological Nomenclature.
Fig. 2: comparison of the stratinomic trace fossil terminology between the systems proposed by Seilacher and Martinsson. |
Whilst this has ameliorated some of the problems faced by ichnologists, the world of trace fossil nomenclature is still somewhat inadequately satisfied in several regards (Pickerill 1994). Some of the difficulties remaining today include: (1) the absence of any real formulated guidelines outlining recommendations with respect to how one should name a newly-discovered trace; (2) the historical "mess" left by early ichnologists who named many ichnotaxa upon the basis of misconception and misidentification (Bromley 1990); and (3) the incredible amount of misspelled ichnotaxa in primary literature and textbooks, among the most common of which is Zoophycus, which even appears in lowercase in Allaby & Allaby 1999 (on the whole, an excellent lexicon).
In addition to the general complicating factors touched upon above, there are many other more specific considerations, which vary in significance depending upon the type of trace. For discussion see Frey 1973, Sarjeant & Kennedy 1973, Häntzschel 1975, Basan 1979, Sarjeant 1979, Bromley 1990, Pickerill 1994, and references therein.
The first trace fossils are very simple trails: The earliest clear indications of animals are trace fossils from about 570 Ma – structures, such as trails, that record animal activity. Before Cambrian time these traces are rare and minute, 1 mm or so wide for the most part, and were probably formed by creeping worms. Younger trace fossils progressively increase in complexity through time.
The beginning of the Cambrian Period at 543 Ma is marked by the first records of larger, vertical traces, and from then until the beginning of the explosive metazoan radiation ~530 Ma, the traces become an order of magnitude wider, more diverse, and more complex, suggesting a gradual diversification of larger animals (Crimes & Droser 1992). Indeed, the lower boundary of the Cambrian is now defined by the occurrence of a distinctive horizontal burrow trace fossil, Trichophycus pedum in the reference section at Fortune Head, southeastern Newfoundland (Fig. 3).
The abrupt appearance of many different kinds of trace fossils at the end of the Proterozoic is regarded as a singular biological event. It represents either the evolutionary origin of soft-bodied metazoans (Cloud 1948; Sepkoski 1978) or the first appearance in the fossil record of common animals capable of leaving preservable traces (Towe 1970; Durham 1978; Runnegar 1982a, b).
Traces are mostly found in marine sediments, but some of the most famous are terrestrial. The earliest evidence we have for for terrestrial activity by animals is provided by trace fossils. Among the oldest are Late Cambrian to no younger than Arenig (Early Ordovician) tracks made by multiple ~50 cm-sized, many legged animals, and preserved in an aeolian sandstone in the Nepean Formation (Potsdam Group) near Kingston, Ontario. However, these track-makers were probably amphibious arthropods - possibly euthycarcinoids - which only left the sea for a limited time, rather than fully terrestrial animals (MacNaughton et al. 2002).
 Fig. 3: The horizontal burrow trace fossil, Trichophycus (formerly Phycodes) pedum defines the lower boundary of the Cambrian in the reference section at Fortune Head, southeastern Newfoundland. [Image courtesy of Dr. Gerd Geyer, Institut für Paläontologie, Bayerische Julius-Maximilians-Universität, Würzburg, Germany.] |
Among larger and more recent animals, dinosaur trackways are relatively common, intensively studied, and have engendered an entire literature of their own. Perhaps the most famous, however, is the hominid trackway discovered by Mary Leakey's team at Laetoli (Fig. 4).
Although individual traces can yield important information about the rocks within which they are found, e.g. U-shaped burrows make excellent geopetal indicators, generally, a single trace fossil alone is a poor indicator of the environment in which the trace maker lived. Trace fossil associations, however, can prove to be extremely useful tools in palaeoenvironmental interpretation. The two most powerful concepts in this regard are ichnocoenoses and ichnofacies, which are quite similar and might easily be confused with each other, so some initial definitions are perhaps in order. An ichnofacies is a rock sequence, the defining characteristics of which include its lithology and sedimentary structures (of which the only lebensspuren considered are specific trace fossils). An ichnocoenosis is an assembly of trace fossils that were all generated by members of the same community (Allaby & Allaby 1999). They are used as components of ichnofacies, comprising less ichnodiversity than the broader range provided by the trace fossil signature of a full rock sequence.
(A)  |
(B)  |
| Fig. 4: Fossil hominid trackway site, known as Laetoli Site G, located in northern Tanzania. One of its trackways records unique evidence of bipedalism in hominids 3.6 million years ago. A team led by Mary Leakey originally excavated the site during 1978-1979. [Image (B) courtesy of University of Liverpool.] | |
Specific types of ichnocoenosis and ichnofacies are given taxonomic names that distinguish them from other types, e.g. the Fuersichnus ichnocoenosis, which falls into the Cruziana ichnofacies (Bromley & Asgaard 1991). The names are supplied by the ichnogenus that is most typically found within the ichnocoenosis or ichnofacies, but this ichnogenus does not necessarily have to be present in each individual occurrence. On the other hand, a certain ichnogenus or ichnocoenosis may not always be restricted to one particular ichnofacies, with many having features that overlap between two different groups. For example, Cruziana ichnospecies are prevalent in the Cruziana ichnofacies, but are also present as part of the Scoyenia ichnofacies, as are Skolithos ichnospecies (which also occur in the Glossifungites ichnofacies).
There has been some confusion as to the true application of ichnofacies, with many authors (e.g. Lockley et al. 1987) suggesting there is a strong palaeobathymetric link. Indeed, this was the manner in which they were first introduced by Seilacher 1963, following his work on trace fossil associations throughout the fifties (e.g. Seilacher 1955). Where ichnocoenoses and ichnofacies occur, however, most attention should be paid to depositional conditions or environmental gradients, with palaeo-bathymetry being a prominent control only where certain environmental parameters are also related to bathymetry (Frey et al. 1990). There is also a stronger taphonomic control than many authors seem to appreciate, with trace assemblages not necessarily accurately reflecting the work of the original community (Bromley 1990). Bromley & Asgaard (1991) argue the importance of taphonomic control with a strong example in that the Nereites and Zoophycos ichnofacies may well reflect identical communities. The Nereites ichnofacies represents shallower tiers of the community, which are often destroyed by the deeper-tiered Zoophycos traces. Therefore "these would appear to be two taphonomic aspects of the same incipient ichnocoenosis, and therefore have the attributes of a taphofacies."
There are several currently recognised ichnofacies, some of which are more poorly defined and established on more tenuous grounds than others. The original four were proposed by Seilacher (1964) as a succession (Skolithos → Cruziana → Zoophycos → Nereites) to aid palaeoenvironmental interpretation from shallow- to deep-water rock sequences respectively. All four are marine ichnofacies produced chiefly by bioturbational behaviour. Upon refinement of Seilacher's original scheme more ichnofacies have been identified, not all restricted to the marine realm, also including those of a bioerosive nature, and the palaeobathymetric link has lost much of its credibility (as discussed above). Seilacher (1967) himself began to argue against the reliability of ichnofacial bathymetric profiling, when he proposed two further ichnofacies, Glossifungites and Scoyenia, both independent of water depth. The Scoyenia ichnofacies has since come under scrutiny, with several authors (e.g. Bromley & Asgaard 1991) highlighting certain problems with it, such as low ichnodiversity and its similarity to other assemblages. The Psilonichnus, Teredolites, and Trypanites ichnofacies make up the rest of the nine "major" currently accepted ichnofacies.
The Cruziana ichnofacies is characteristic of a low-energy environment, best developed in circalittoral substrates below a normal fair-weather wave base (but not below the storm wave base) in well-sorted sands and silts (Brenchley & Harper 1998, Frey et al. 1990). Such quiet conditions might be expected in estuarine, lagoonal, and shelf environments, as well as certain non-marine scenarios, where lower ichnodiversity occurs. The traces are made up of burrows created by suspension and detritus feeders, and most prominently, repichnia reflecting the activities of mobile carnivores (the facies is named after a crawling trilobite trace).
The Glossifungites ichnofacies was erected by Seilacher 1967 to accommodate firm, often exhumed substrates such as dewatered muds (e.g. Brenchley & Harper, 1998). It occurs in conditions of moderate energy or in high-energy settings where semi-consolidated micritic/siliciclastic substrates offer some resistance to erosion (Frey et al. 1990). It has been recognised as a transitional phase of benthic community succession, as a hardground develops from an original softground in the natural evolution of an omission surface (Goldring & Kazmierczak 1974, Fürsich 1978), and therefore has little bathymetric restriction. Borings prevail in this predominantly bioerosive facies, along with burrows, some of which are quite complex (e.g. Spongeliomorpha Saporta 1887).
One of the original ichnofacies proposed by Seilacher 1964 as a set of recurring, palimpsest ichnofossil assemblages, the Nereites ichnofacies is thought to develop on and in quiet but moderately well-oxygenated seabeds (Brenchley & Harper 1998). The palaeodepth is usually from bathyal to abyssal, and it is common in flysch sequences where the area has been disrupted by down-canyon bottom currents and turbidity flows (Frey et al. 1990). A wide diversity of agrichnia, pascichnia, and repichnia is seen, the bioturbation taking the form of complex structures that are allowed to form in slowly-accreting, distal abyssal conditions (Frey et al. 1990). Seilacher 1964 identifies two distinct suites: (1) pre-turbidite assemblages, developing in mud softgrounds and representing the topmost and shallowest tiers of benthic activity; and (2) post-turbidite assemblages, occurring in and on sand softgrounds in the form of flysch systems. Some authors (e.g. Bromley & Asgaard 1991) hold only the pre-turbidite suite to truly represent the ichnofacies.
The Psilonichnus ichnofacies was proposed by Frey & Pemberton (1987) and based on traces associated with well-sorted, variably laminated to cross-stratified sands, to root- and burrow-mottled, poorly-sorted muddy sands. This implies moderate- to low-energy marine and/or aeolian conditions, such as sand dunes in both beach and backshore environments, and the coastal setting may extend to washover fans and supratidal flats (Frey et al. 1990).
Introduced by Seilacher 1967, and perhaps the most debated of the nine major ichnofacies, Scoyenia is characteristic of terrestrial redbed sequences (e.g. Brenchley & Harper 1998). These are formed in conditions intermediate between fully aquatic and non-aquatic, non-marine environments, at low-energy sites. The moist to wet sandy sediments may also support semi-aquatic vegetation (Frey et al. 1990).
Perhaps one of the best-known ichnofacies, and indeed ichnogenera, is Skolithos. The trace itself is a simple, straight, vertical burrow with a circular cross-section, created by suspension-feeding animals. The ichnofacies is marine, representing moderate- to relatively high-energy conditions, such as a beach foreshore or shoreface, other possible settings including estuarine point bars, tidal deltas, and deep-sea sand fans (Frey et al. 1990). The trace fossil signature of the facies is, as one might expect, on the pattern of Skolithos-type burrows, such as Diplocraterion and Monocraterion Torell 1870. Such burrows are often abundant, but the ichnodiversity is usually low.
The Teredolites ichnofacies is unique in developing in xylic skeletal material as wood-boring structures (Bromley et al. 1984). It represents predominantly a marine environment, where the borings are produced chiefly by bivalves, but freshwater ichnocoenoses can occur, in which case the borings consist primarily of those made by isopods (Frey et al. 1990).
The bioerosive Trypanites ichnofacies reflects production in hard substrates, such as hardgrounds and rockgrounds (Frey & Seilacher 1980). Typical casting media include omission surfaces on rocky coasts, beach rock, carbonates, and organic substrates such as reefs and shell beds (Frey et al. 1990). Originally described as marine, it can also occur in non-marine conditions, being produced on rigid skeletal material. This property, together with the occurrence of hardgrounds and rockgrounds at all water depths, gives the Trypanites ichnofacies perhaps the widest distribution of all ichnofacies (Bromley & Asgaard 1991). Because of the sequentiality of hardground development, the Trypanites suites may be seen to cross-cut previously formed ichnofacies such as Glossifungites, or even earlier assemblages attributable to the Cruziana ichnofacies (Frey et al. 1990).
Seilacher 1964 introduced the Zoophycos ichnofacies as one requiring quiet water and (presumably) nutrient-rich conditions on the outer shelf and slope. It can also develop in both shallower and deeper environments where similar conditions prevail (e.g. a lagoonal environment or a deep-sea setting supplied by rich down-welling nutriment). It is dominated by complex fodinichnia and pascichnia created by deposit feeders. There is some debate as to the degree of oxygenation on the seafloor represented here, with many authors (e.g. Frey & Pemberton 1984; Frey et al. 1990) suggesting a deficiency. Others (e.g. Bromley & Ekdale 1984, Bromley & Asgaard 1991) propose that this is not the case, the organisms being able to burrow to reach their required reducing pore-water environments.
Several authors have proposed additions and/or augmentations to the current ennead of major ichnofacies. For example, Bromley & Asgaard 1991 suggests the dissolution of the Scoyenia ichnofacies and inclusion of the Arenicolites ichnofacies instead, and the same two authors in 1993 divided the Trypanites ichnofacies into the Gnathichnus and Entobia ichnofacies. It has been suggested that the Nereites ichnofacies should be split into the Nereites and Palaeodictyon ichnofacies (Brenchley & Harper 1998), because of the apparent mutual exclusivity of some of the trails and burrows, and there are many more examples. Other less important ichnofacies are generally non-marine in nature and include, among others, the lacustrine Mermia (Buatois & Mángano 1995) and palaeosolic Coprinisphaera (Genise et al. 2000) ichnofacies. This section has focussed on the palaeoenvironmental implications of the nine ichnofacies that have been most prominent since the proposal of Seilacher's original four in 1964.
The basic fact is that, despite their usefulness, ichnofacies are by no means absolutely consistent; variations will always be observed. If one were to base an ichnofacies in any rigorously specific way then a potentially indefinite number would be discovered, due to local variations in (e.g.) ecology, taphonomy, and diagenetic/depositional controls. It is thought, however, that many more terrestrial ichnofacies are yet to be recognised and defined (Buatois & Mángano 1995). As far as current ichnological understanding allows, ichnofacies provide a reasonable proxy for palaeoenvironments.
Links: Emory University (Atlanta, Georgia) – Introduction to Ichnology; University of Arizona site
Allaby, A.; Allaby, M. 1999: Oxford Dictionary of Earth Sciences. Oxford University Press, London.
Andre 1920
Basan, P.B. 1979: Trace fossil nomenclature: the developing picture. Palaeogeography, Palaeoclimatology, Palaeoecology, 28: 143-6.
Benton, M.J.; Gray, D.I. 1981: Lower Silurian distal shelf storm-induced deposits in the Welsh Borders: sediments, tool marks and trace fossils. Journal of the Geological Society of London, 138: 675-94.
Bown 1982
Bown, T.M.; Ratcliffe, B.C. 1988: The origin of Chubutolithes Ihering, ichnofossils from the Eocene and Oligocene of Chubut province, Argentina. Journal of Paleontology 62: 163-167.
Brenchley, P.J.; Harper, D.A.T. 1998: Palaeoecology: Ecosystems, environments and evolution. Chapman & Hall, London, 402 pp.
Bromley, R.G. 1981: Concepts in ichnotaxonomy illustrated by small round holes in shells. Acta Geologica Hispanica 16: 55-64.
Bromley, R.G. 1990: Trace fossils: biology and taphonomy. Unwin Hyman Ltd, London, 280 pp.
Bromley, R.G. 1994: The palaeoecology of bioerosion. In Donovan, S.K. (ed.) 1994: The paleobiology of trace fossils. John Wiley & Sons, Chichester: 134-154.
Bromley, R.G. 1996: Trace Fossils: biology, taphonomy and applications (2nd edition). Chapman & Hall, London, 361 pp.
Bromley, R.G.; Asgaard, U. 1991: Ichnofacies: a mixture of taphofacies and biofacies. Lethaia 24: 153-163.
Bromley, R.G.; Asgaard, U. 1993: Endolithic community replacement on a Pliocene rocky coast. Ichnos 2: 93-116.
Bromley, R.G.; Ekdale, A.A. 1984: Chondrites: a trace fossil indicator of anoxia in sediments. Science 224: 872-874.
Bromley, R.G.; Pemberton, S.G.; Rahmani, R.A. 1984: A Cretaceous woodground: the Teredolites ichnofacies. Journal of Paleontology 58: 488-498.
Buatois, L.A.; Mángano, M.G. 1995: The paleoenvironmental and paleoecological significance of the lacustrine Mermia ichnofacies: an archetypical subaqueous nonmarine trace fossil assemblage. Ichnos 4: 151-161.
Chamberlain, C.K. 1971: Morphology and ethology of trace fossils from the Ouachita Mountains, southeast Oklahoma. Journal of Paleontology, 45: 212-246.
Cloud, P.E. 1948: Some problems and patterns of evolution exemplified by fossil invertebrates. Evolution 2(4): 322-350.
Crimes, T.P.; Droser, M.L. 1992: Trace fossils and bioturbation: The other fossil record. Annual Reviews of Ecology and Systematics 23: 339-360.
Donovan, S.K. 1994: The palaeobiology of trace fossils. John Wiley & Sons Ltd, 308 pp.
d'Orbigny 1842
Durham, J.W. 1978: A Lower Cambrian eocrinoid. Journal of Paleontology 52: 195-199.
Ehrenberg 1944
Ekdale, A.A. 1985: Palaeoecology of the marine endobenthos. Palaeogeography, Palaeoecology, Palaeoclimatology 50: 63-81.
Ekdale, A.A.; Bromley, R.G.; Pemberton, S.G. 1984: Ichnology: Trace fossils in sedimentology and stratigraphy. Society of Economic Paleontologists and Mineralogists Short Course, no. 15, 317 pp.
Frey, R.W. 1973: Concepts in the study of biogenic sedimentary structures. Journal of Sedimentary Petrology 43: 6-19.
Frey, R.W.; Pemberton, S.G. 1987: The Psilonichnus ichnocoenose, and its relationship to adjacent marine and nonmarine ichnocoenoses along the Georgia coast. Bulletin of Canadian Petroleum Geology 35: 333-357.
Frey, R.W.; Pemberton, S.G.; Saunders, T.D.A. 1990: Ichnofacies and bathymetry: a passive relationship. Journal of Paleontology 64: 155-158.
Frey, R.W.; Seilacher, A. 1980: Uniformity in marine invertebrate ichnology. Lethaia 13: 183-207.
Fuchs 1895
Fürsich 1974
Fürsich, F.T. 1978: The influence of faunal condensation and mixing on the preservation of fossil benthic communities. Lethaia 11: 243-250.
Geinitz 1849
Genise, J.; Bown, T.M. 1994
Genise, J.F.; Mángano, M.G.; Buatois, L.A.; Laza, J.H.; Verde, M. 2000: Insect trace fossil associations in paleosols: the Coprinisphaera ichnofacies. Palaios 15: 49-64.
Gibert, J.M. de; Domènech, R.; Martinell, J. 2004: An ethological framework for animal bioerosion trace fossils upon mineral substrates with proposal of new class, fixichnia. Lethaia 37 (4): 429-437.
Goldring, R.; Kazmierczak, J. 1974: Ecological succession in intraformational hardground formation. Palaeontology 17: 949-962.
Haldeman 1840
Hall 1852
Häntzschel, W. 1975: Trace fossils and problematica. In Teichert, C. (ed.) 1975: Treatise of Invertebrate Paleontology (2nd Edition), part W, Miscellanea, Supp 1. University of Kansas and Geological Society of America, Boulder, Colorado, and Lawrence, Kansas, 269 pp.
Hasiotis, S.T. 2003: Complex ichnofossils of solitary and social soil organisms: understanding their evolution and roles in terrestrial paleoecosystems. Palaeogeography, Palaeoclimatology, Palaeoecology 192 (1-4): 259-320.
Hitchcock, E. 1848
Ihering 1922
Lockley, M.G. Rindsberg, A.K.; Zeiler, R.M. 1987: The paleoenvironmental significance of the nearshore Curvolithus ichnofacies. Palaios 2: 255-262.
| Lagerstätten |
page by Kristian Saether & Christopher Clowes - original url
edited RFVS111204